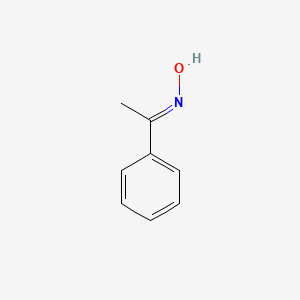
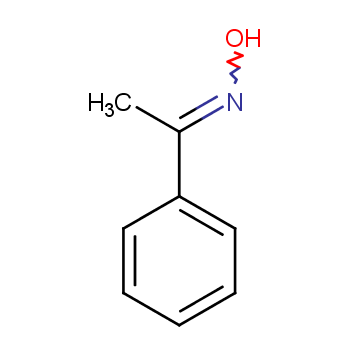
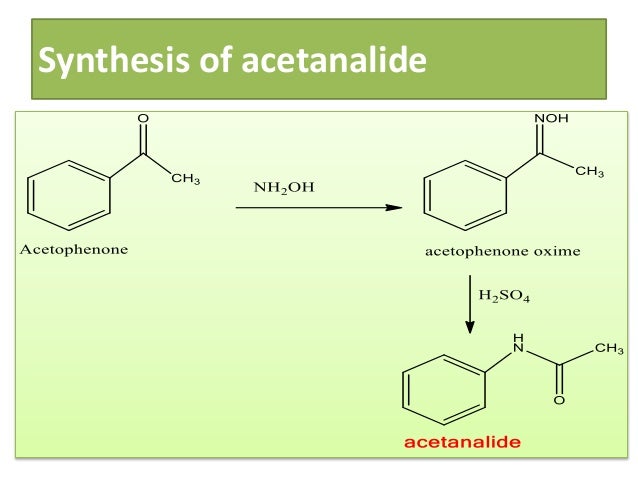
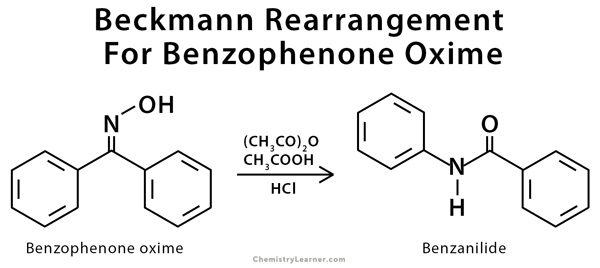
Beckmann rearrangement of acetophenone oximes to the corresponding amides organo-catalyzed by trifluoroacetic acid for sustainable NSAIDs synthesis - ScienceDirect
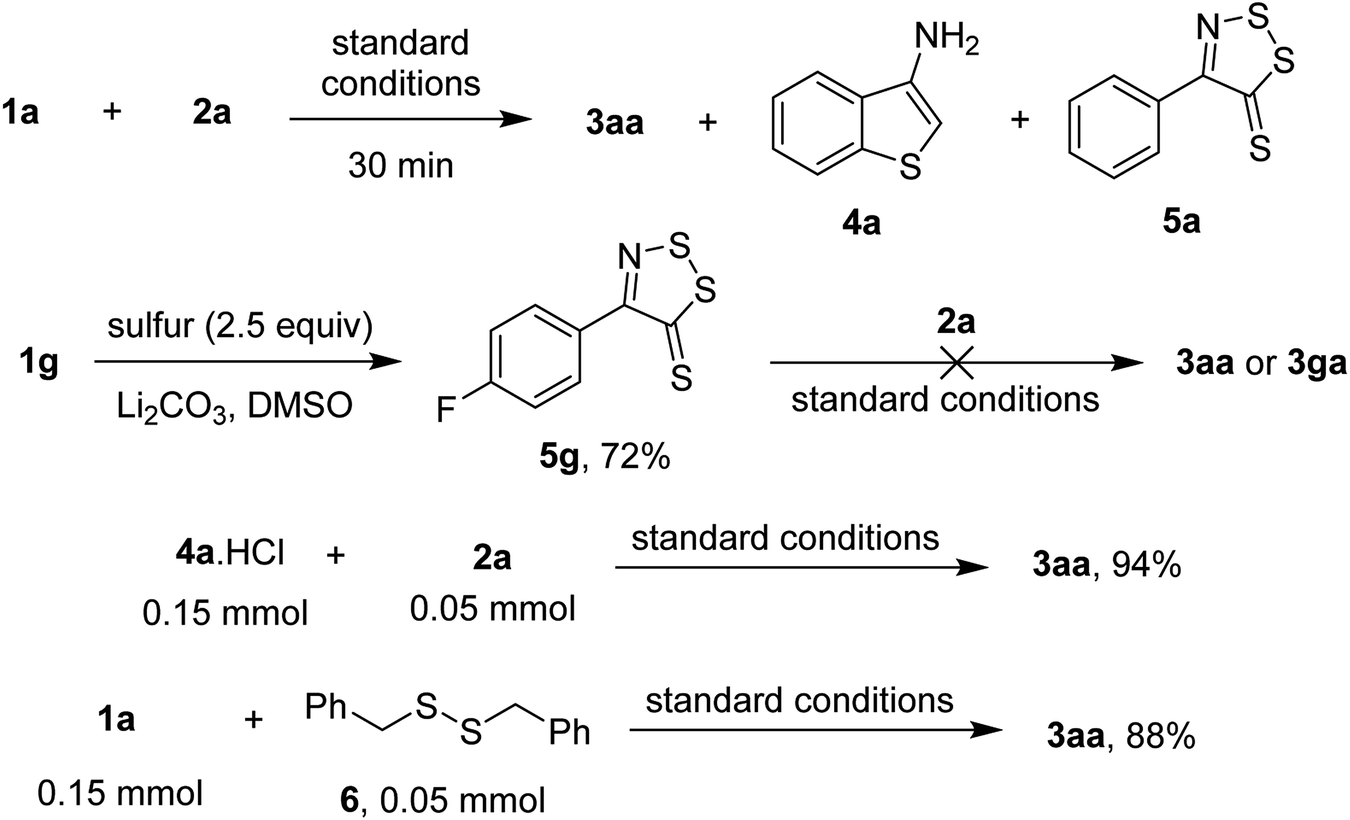
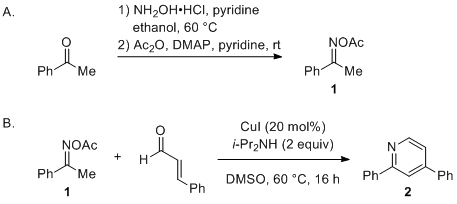
Functionalization of C–H bonds in acetophenone oximes with arylacetic acids and elemental sulfur - RSC Advances (RSC Publishing)

Figure 6 from Synthesis of 3-( 1 H-1-imidazolyl )-2-methyl-4-chromanone as a pharmaceutical composition default by α-bromo-2-hydroxy acetophenone | Semantic Scholar

Beckmann rearrangement of acetophenone oximes to the corresponding amides organo-catalyzed by trifluoroacetic acid for sustainable NSAIDs synthesis - ScienceDirect
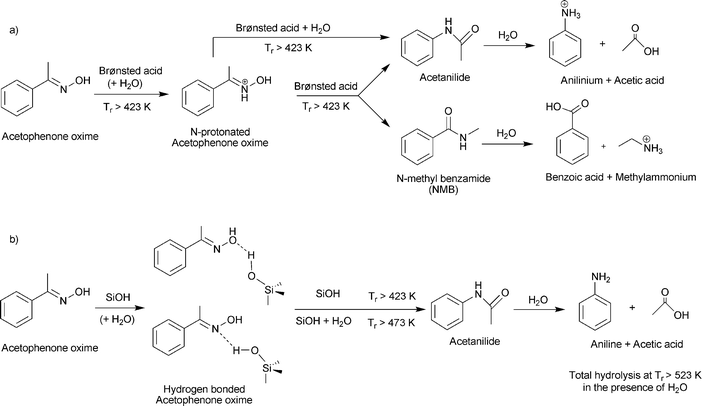
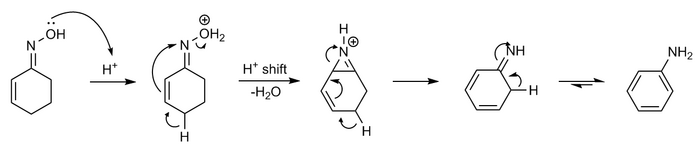
Study of the Beckmann rearrangement of acetophenone oxime over porous solids by means of solid state NMR spectroscopy - Physical Chemistry Chemical Physics (RSC Publishing)
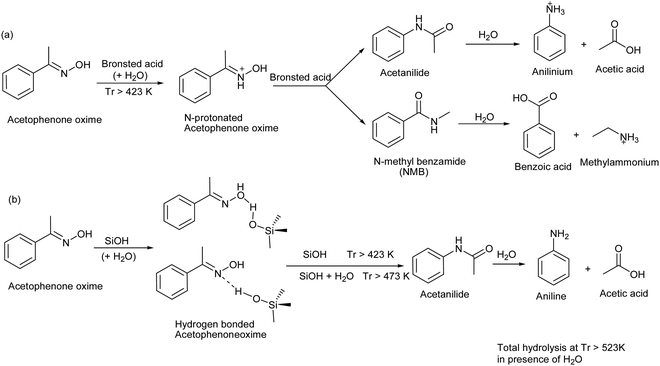
Beckmann rearrangement catalysis: a review of recent advances - New Journal of Chemistry (RSC Publishing) DOI:10.1039/D0NJ02034F

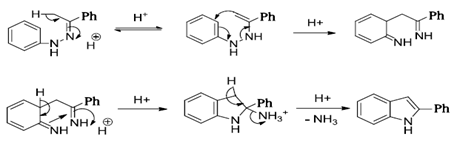
Chemical Sciences: A Manual for CSIR-UGC National Eligibility Test for Lectureship and JRF/Named Reactions/Beckmann Rearrangement - Wikibooks, open books for an open world

PDF) Synthesis of Some Acetophenone Oximes and Their Corresponding Bridged Terphthaloyl Oxime Esters

Orthopalladated acetophenone oxime compounds bearing thioamides as ligands: Synthesis, structure and cytotoxic evaluation - ScienceDirect
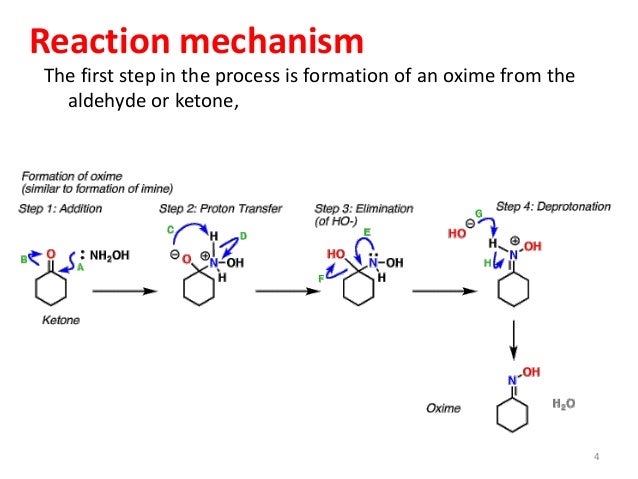
Oxime formation from hydroxylamine and ketone: a (computational) reality check on stage one of the mechanism. | Henry Rzepa's Blog
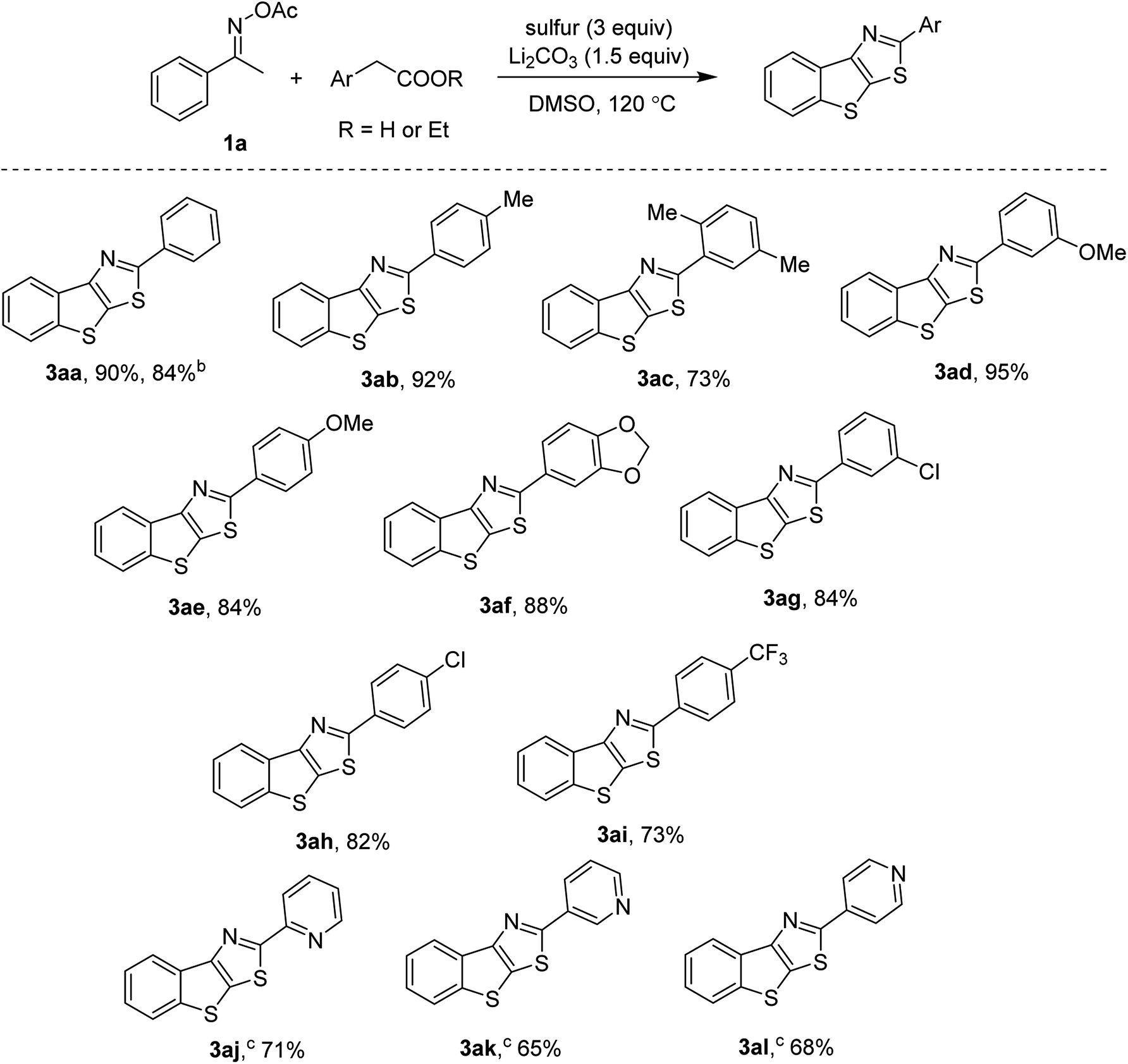
Functionalization of C–H bonds in acetophenone oximes with arylacetic acids and elemental sulfur - RSC Advances (RSC Publishing)