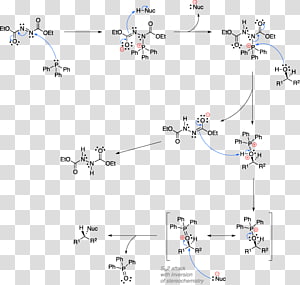
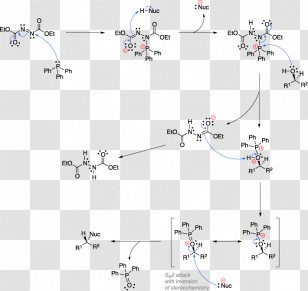
The Curtius rearrangement: mechanistic insight and recent applications in natural product syntheses. - Abstract - Europe PMC
![Diphenylphosphoryl Azide (DPPA)‐Mediated One‐Pot Synthesis of Oxazolo[4,5‐c][1,8]naphthyridin‐4(5 H)‐ones, Oxazolo[4,5‐c]quinoline‐4(5 H)‐ones, and Tosyloxazol‐5‐yl Pyridines - Tangella - 2017 - Asian Journal of Organic Chemistry - Wiley Online Library Diphenylphosphoryl Azide (DPPA)‐Mediated One‐Pot Synthesis of Oxazolo[4,5‐c][1,8]naphthyridin‐4(5 H)‐ones, Oxazolo[4,5‐c]quinoline‐4(5 H)‐ones, and Tosyloxazol‐5‐yl Pyridines - Tangella - 2017 - Asian Journal of Organic Chemistry - Wiley Online Library](https://onlinelibrary.wiley.com/cms/asset/103f5bd3-e4c8-4a4d-9117-372733c08994/ajoc201700147-fig-5005-m.jpg)
Diphenylphosphoryl Azide (DPPA)‐Mediated One‐Pot Synthesis of Oxazolo[4,5‐c][1,8]naphthyridin‐4(5 H)‐ones, Oxazolo[4,5‐c]quinoline‐4(5 H)‐ones, and Tosyloxazol‐5‐yl Pyridines - Tangella - 2017 - Asian Journal of Organic Chemistry - Wiley Online Library
![Diphenylphosphoryl Azide (DPPA)‐Mediated One‐Pot Synthesis of Oxazolo[4,5‐c][1,8]naphthyridin‐4(5 H)‐ones, Oxazolo[4,5‐c]quinoline‐4(5 H)‐ones, and Tosyloxazol‐5‐yl Pyridines - Tangella - 2017 - Asian Journal of Organic Chemistry - Wiley Online Library Diphenylphosphoryl Azide (DPPA)‐Mediated One‐Pot Synthesis of Oxazolo[4,5‐c][1,8]naphthyridin‐4(5 H)‐ones, Oxazolo[4,5‐c]quinoline‐4(5 H)‐ones, and Tosyloxazol‐5‐yl Pyridines - Tangella - 2017 - Asian Journal of Organic Chemistry - Wiley Online Library](https://onlinelibrary.wiley.com/cms/asset/956f3c23-c762-486a-8910-701e6cbe6593/ajoc201700147-fig-5001-m.jpg)
Diphenylphosphoryl Azide (DPPA)‐Mediated One‐Pot Synthesis of Oxazolo[4,5‐c][1,8]naphthyridin‐4(5 H)‐ones, Oxazolo[4,5‐c]quinoline‐4(5 H)‐ones, and Tosyloxazol‐5‐yl Pyridines - Tangella - 2017 - Asian Journal of Organic Chemistry - Wiley Online Library

The Curtius rearrangement: mechanistic insight and recent applications in natural product syntheses. - Abstract - Europe PMC
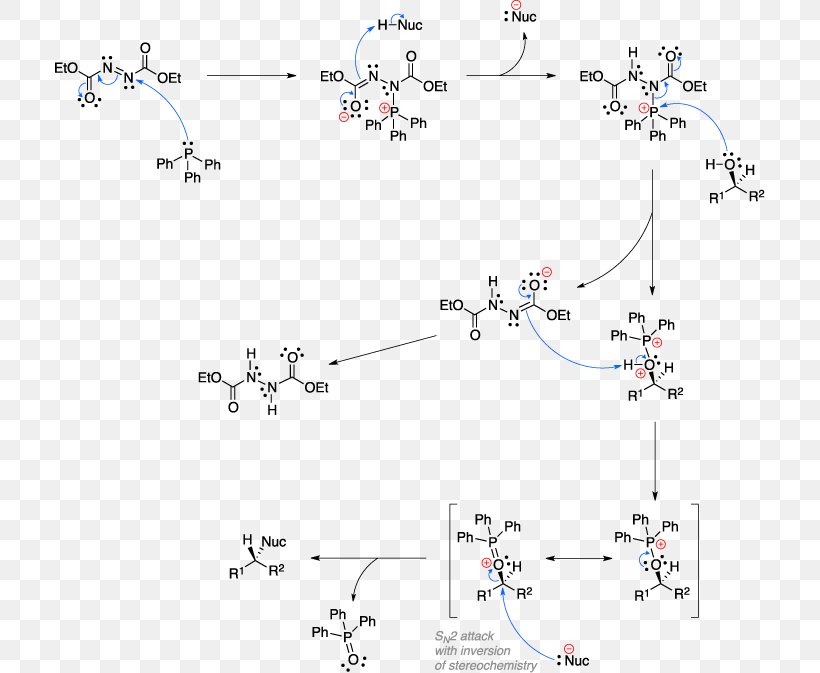
Mitsunobu Reaction Diphenylphosphoryl Azide Chemical Reaction Reaction Mechanism Appel Reaction, PNG, 710x673px, Mitsunobu Reaction, Alcohol, Appel

In situ generation of N-unsubstituted imines from alkyl azides and their applications for imine transfer via copper catalysis
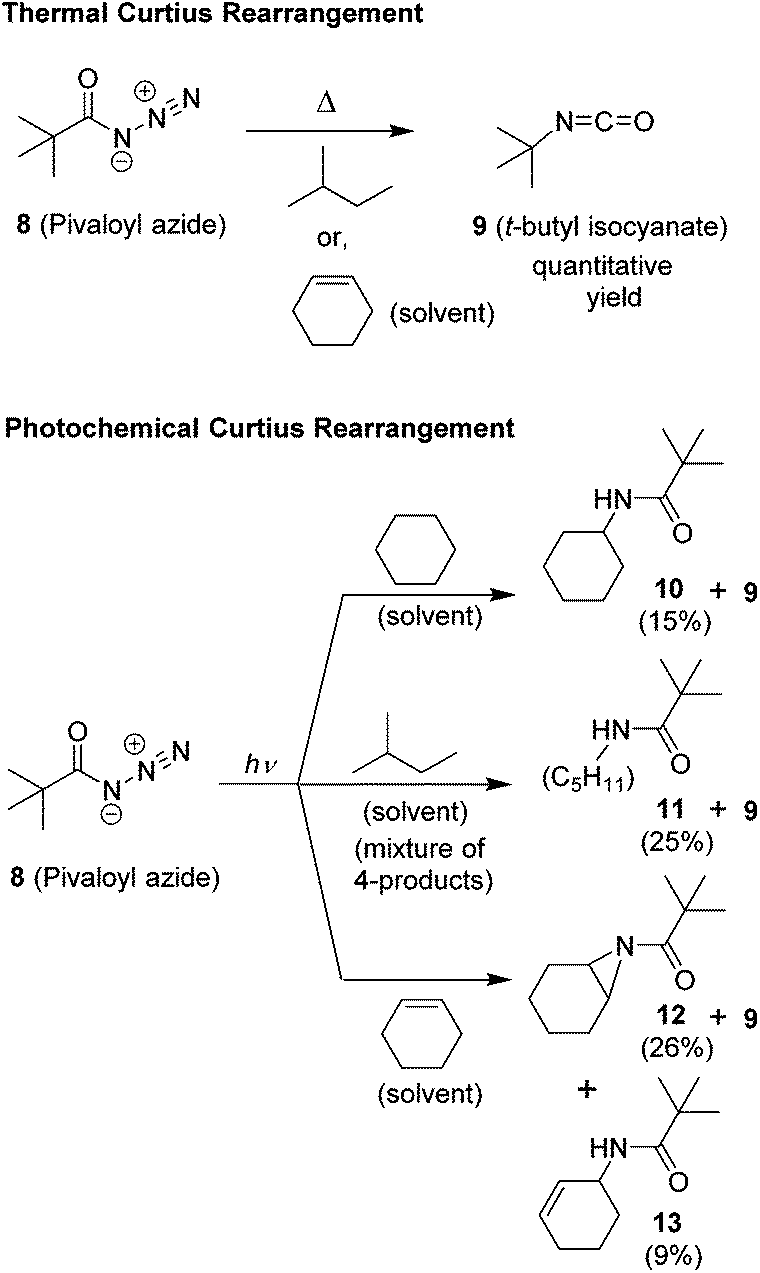
The Curtius rearrangement: mechanistic insight and recent applications in natural product syntheses - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C8OB00138C

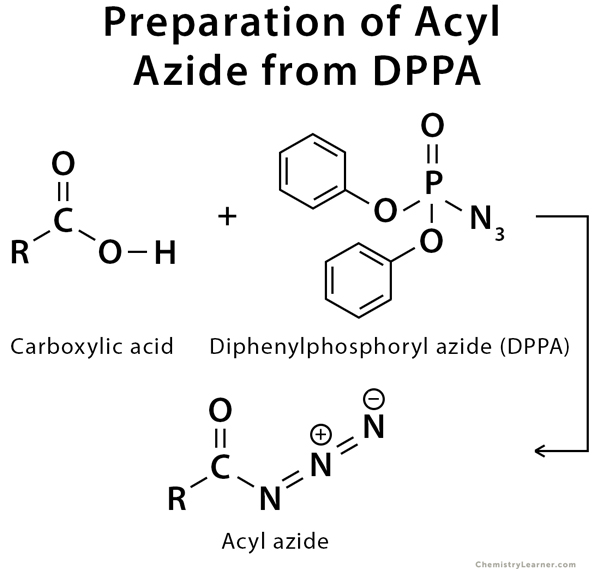
Copper catalyzed synthesis of aryl/alkyl mixed phosphates from diphenylphosphoryl azides and aliphatic alcohols under mild conditions - ScienceDirect

The Curtius Rearrangement: Applications in Modern Drug Discovery and Medicinal Chemistry - Ghosh - 2018 - ChemMedChem - Wiley Online Library
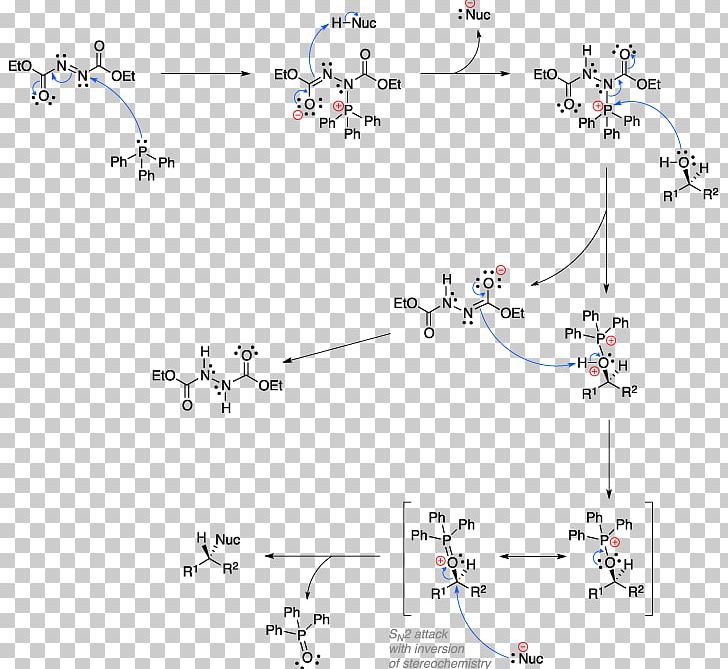
Mitsunobu Reaction Diphenylphosphoryl Azide Chemical Reaction Reaction Mechanism Appel Reaction PNG, Clipart, Alcohol, Appel Reaction, Are,

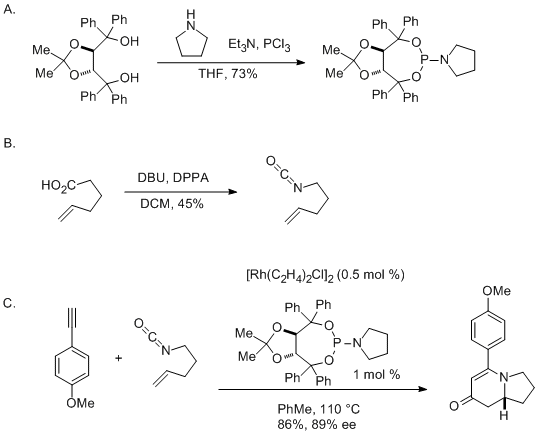
Molecules | Free Full-Text | Development of a Telescoped Flow Process for the Safe and Effective Generation of Propargylic Amines | HTML