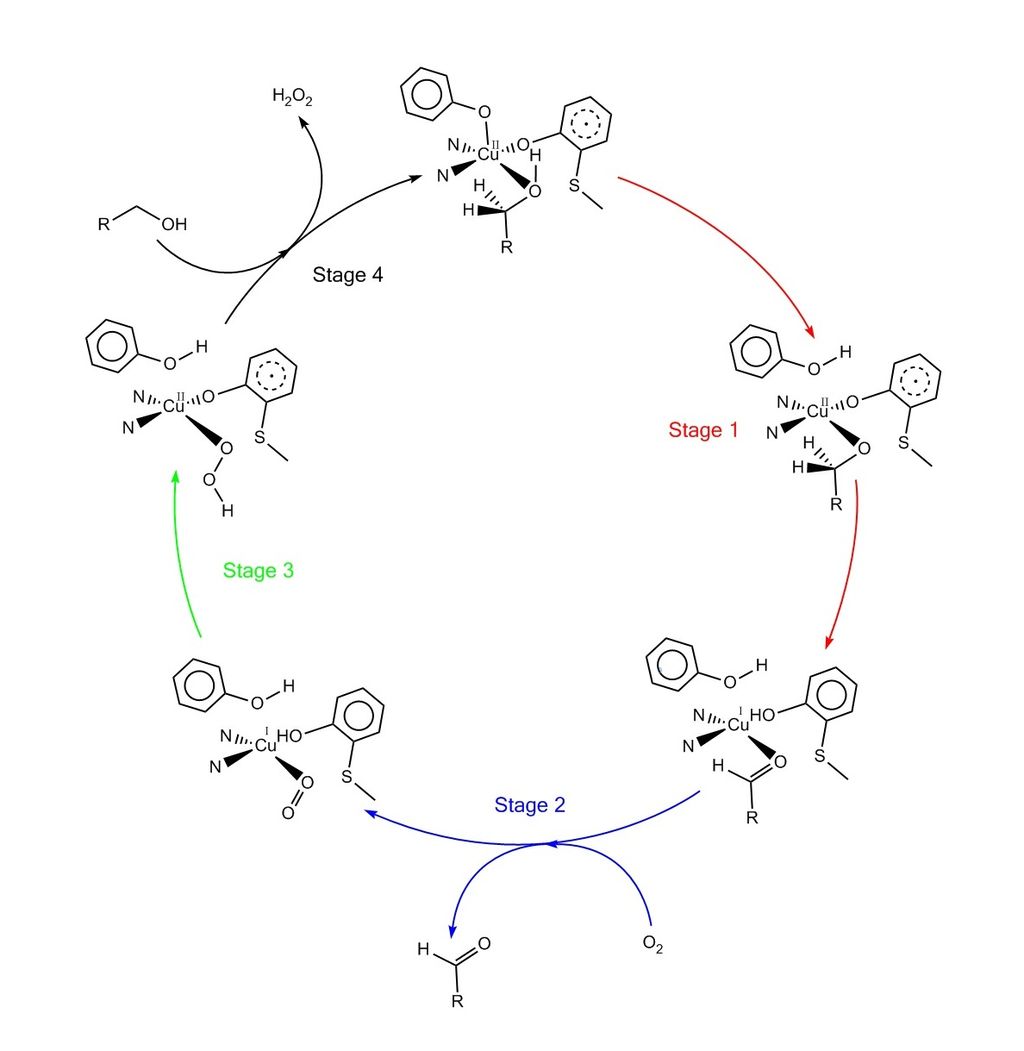
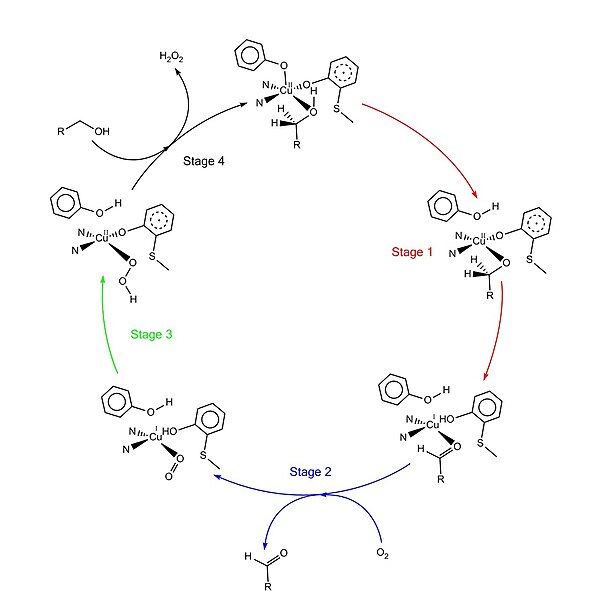
Recent Advances in One‐Electron‐Oxidized CuII–Diphenoxide Complexes as Models of Galactose Oxidase: Importance of the Structural Flexibility in the Active Site - Oshita - 2020 - Chemistry – A European Journal - Wiley Online Library
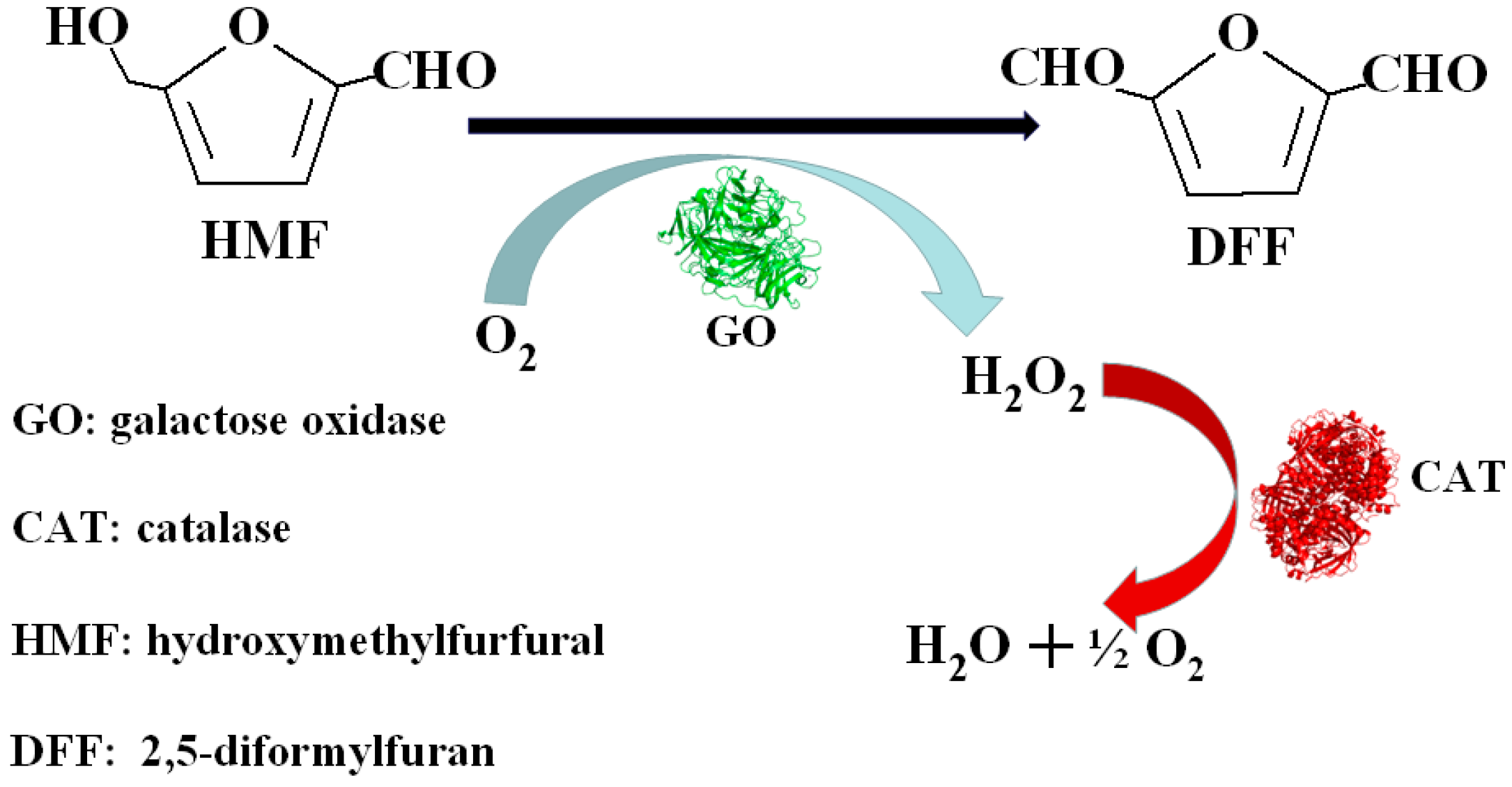
Molecules | Free Full-Text | Co-Immobilization of Tri-Enzymes for the Conversion of Hydroxymethylfurfural to 2,5-Diformylfuran | HTML
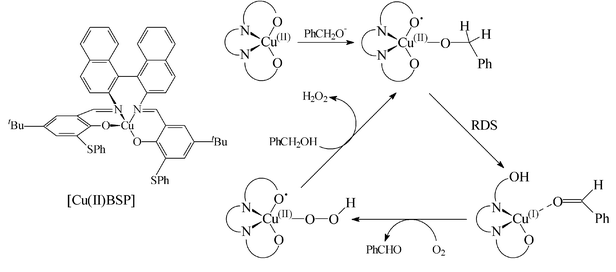
Cu( ii )-nitroxyl radicals as catalytic galactose oxidase mimics - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/B305941C

Cu(I)-dependent Biogenesis of the Galactose Oxidase Redox Cofactor* - Journal of Biological Chemistry

Catalytic Promiscuity of Galactose Oxidase: A Mild Synthesis of Nitriles from Alcohols, Air, and Ammonia - Vilím - 2018 - Angewandte Chemie - Wiley Online Library

Aerobic oxidation of alcohol by model complexes relevant to metal site galactose oxidase: role of copper(I) intermediate, evidence for the generation of end-on copper(II)–OOH species and catalytic promiscuity for oxidation of benzyl