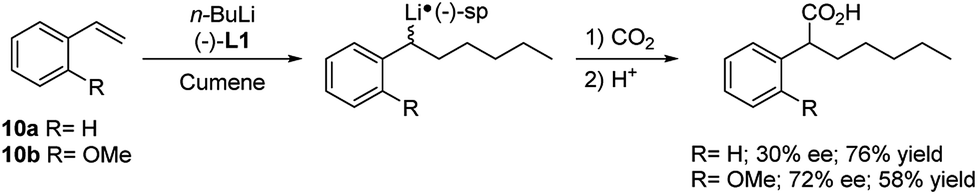
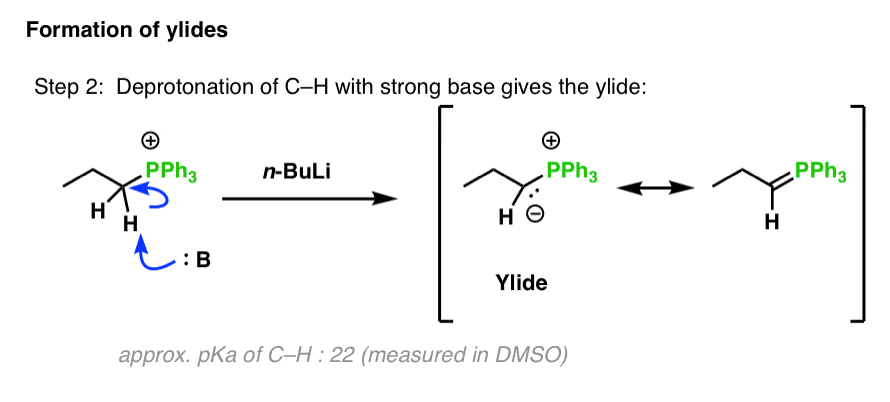
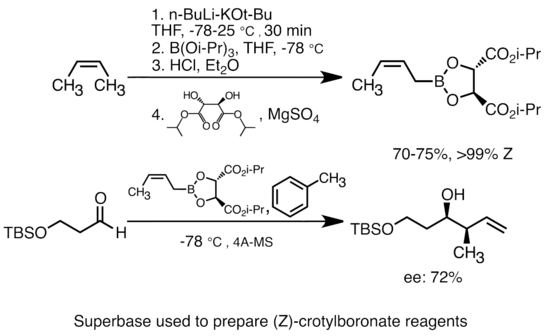
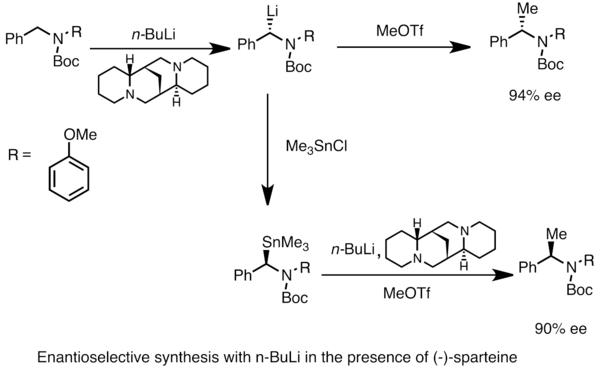
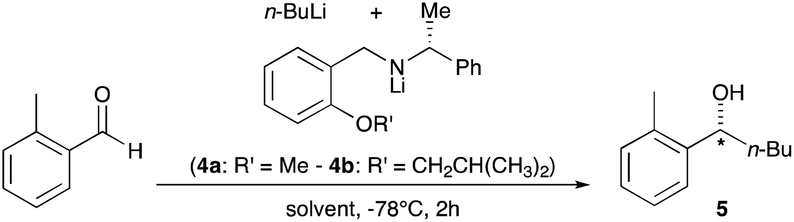
Mechanism of the Deprotonation Reaction of Alkyl Benzyl Ethers with n‐ Butyllithium - Raposo - 2013 - Chemistry – A European Journal - Wiley Online Library
Probing solvent effects on mixed aggregates associating a chiral lithium amide and n -BuLi by NMR: from structure to reactivity - Dalton Transactions (RSC Publishing) DOI:10.1039/C4DT01156B

WO2009056487A1 - Methods for stabilizing lithiated halogen-substituted aromatic compounds - Google Patents
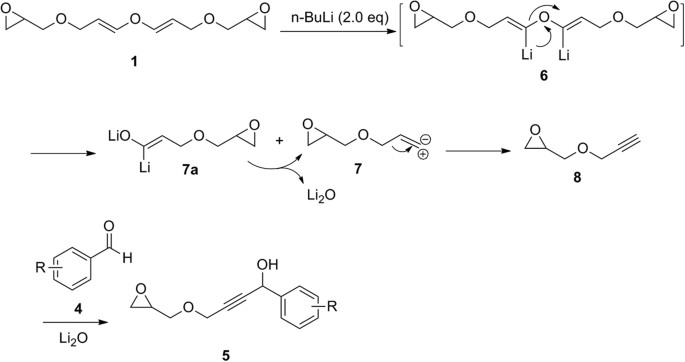
Unprecedented reactions: from epichlorohydrin to epoxyglycidyl substituted divinyl ether and its conversion into epoxyglycidyl propargyl ether | Scientific Reports

Scheme 1 o-Phenylthiostyrene oxide (R)-4 was deprotonated at 157 K with... | Download Scientific Diagram