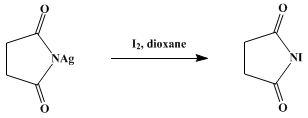![NIS from NCS/NaI: Synthesis of alpha-Iodo Carbonyl Compounds and trans-1,2-Iodoacetates - [www.rhodium.ws] NIS from NCS/NaI: Synthesis of alpha-Iodo Carbonyl Compounds and trans-1,2-Iodoacetates - [www.rhodium.ws]](https://chemistry.mdma.ch/hiveboard/rhodium/pictures/nis.acetoxyiodination.gif)
NIS from NCS/NaI: Synthesis of alpha-Iodo Carbonyl Compounds and trans-1,2-Iodoacetates - [www.rhodium.ws]
![PDF] Highly Regioselective Iodination of Arenes via Iron(III)-Catalyzed Activation of N-Iodosuccinimide. | Semantic Scholar PDF] Highly Regioselective Iodination of Arenes via Iron(III)-Catalyzed Activation of N-Iodosuccinimide. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/0c6f0edeacfadccc931009ae2c66114570d95279/2-Table2-1.png)
PDF] Highly Regioselective Iodination of Arenes via Iron(III)-Catalyzed Activation of N-Iodosuccinimide. | Semantic Scholar

Photoinduced cyclization of alkynoates to coumarins with N-Iodosuccinimide as a free-radical initiator under ambient and metal-free conditions - ScienceDirect

N‐Iodosuccinimide (NIS) in Direct Aromatic Iodination - Bergström - 2017 - European Journal of Organic Chemistry - Wiley Online Library
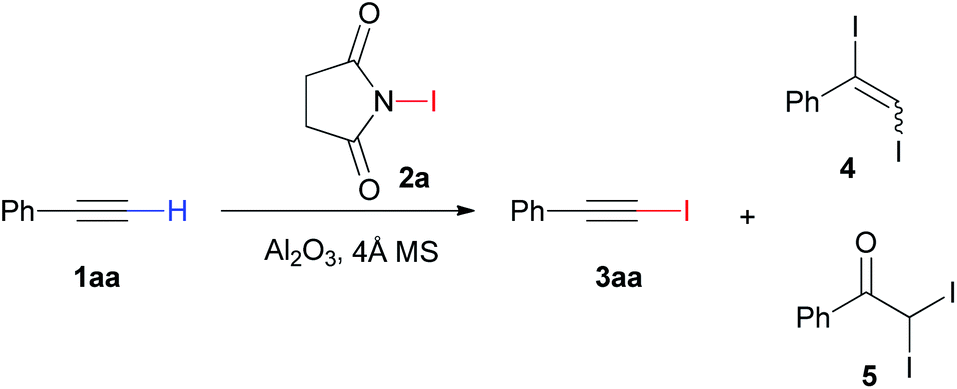
Efficient synthesis of 1-iodoalkynes via Al 2 O 3 mediated reaction of terminal alkynes and N -iodosuccinimide - RSC Advances (RSC Publishing) DOI:10.1039/D0RA00251H

N‐Iodosuccinimide (NIS) in Direct Aromatic Iodination - Bergström - 2017 - European Journal of Organic Chemistry - Wiley Online Library

N‐Iodosuccinimide as Bifunctional Reagent in (E)‐Selective C(sp2)−H Sulfonylation of Styrenes - Pramanik - 2019 - Asian Journal of Organic Chemistry - Wiley Online Library
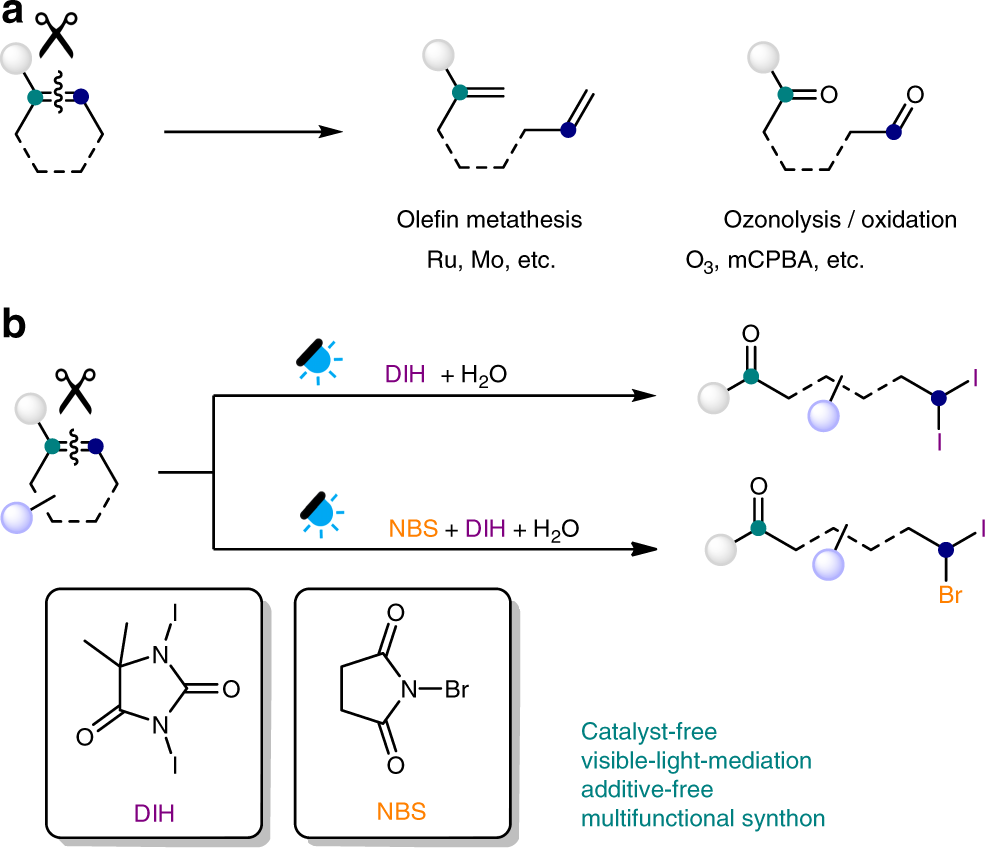
Photo-mediated selective deconstructive geminal dihalogenation of trisubstituted alkenes | Nature Communications
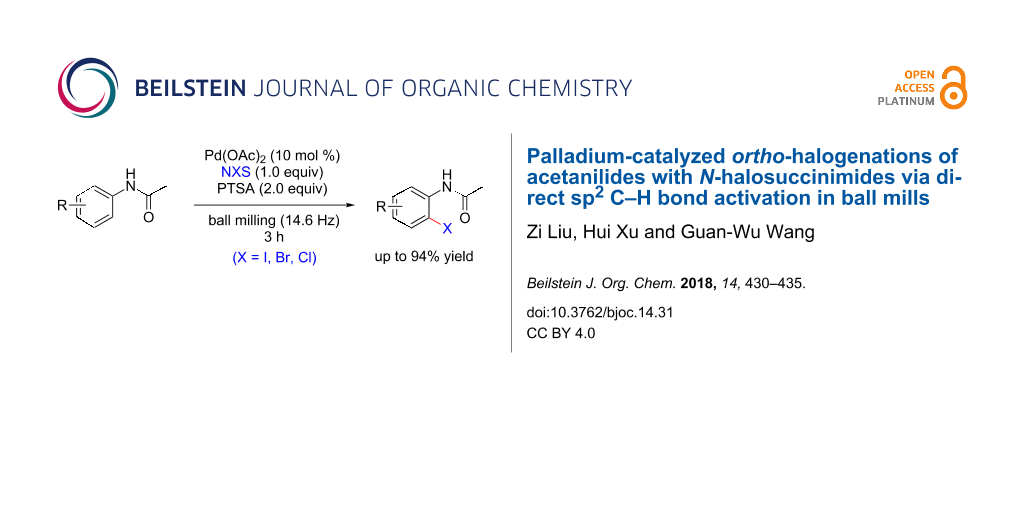
BJOC - Palladium-catalyzed ortho-halogenations of acetanilides with N-halosuccinimides via direct sp2 C–H bond activation in ball mills

N-Iodosuccinimide-Promoted Cascade Trifunctionalization of Alkynoates: Access to 1,1-Diiodoalkenes | Organic Letters
![Chemoselective iodination of 6-substituted imidazo[1,2-a]pyridine | SpringerLink Chemoselective iodination of 6-substituted imidazo[1,2-a]pyridine | SpringerLink](https://media.springernature.com/lw685/springer-static/image/art%3A10.1007%2Fs10593-018-2307-x/MediaObjects/10593_2018_2307_Figa_HTML.gif)





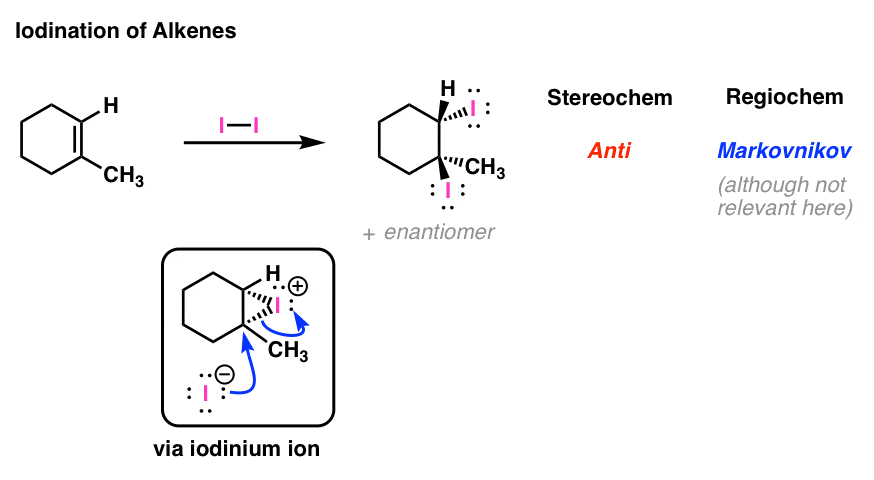
![Iodination [Synthetic Reagents] | Tokyo Chemical Industry Co., Ltd.(APAC) Iodination [Synthetic Reagents] | Tokyo Chemical Industry Co., Ltd.(APAC)](https://www.tcichemicals.com/assets/cms-images/01047.png)