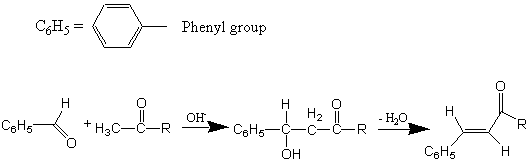Write a reaction that would take place during the synthesis of dibenzalacetone if the concentration of acetone in the reaction mixture is not carefully controlled. | Study.com
OneClass: Lab: Synthesis of Dibenzalacetone---An aldol condensation a) Write the balanced equation fo...
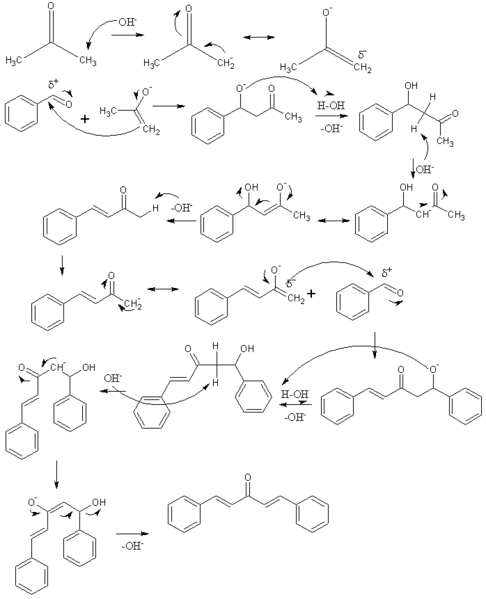
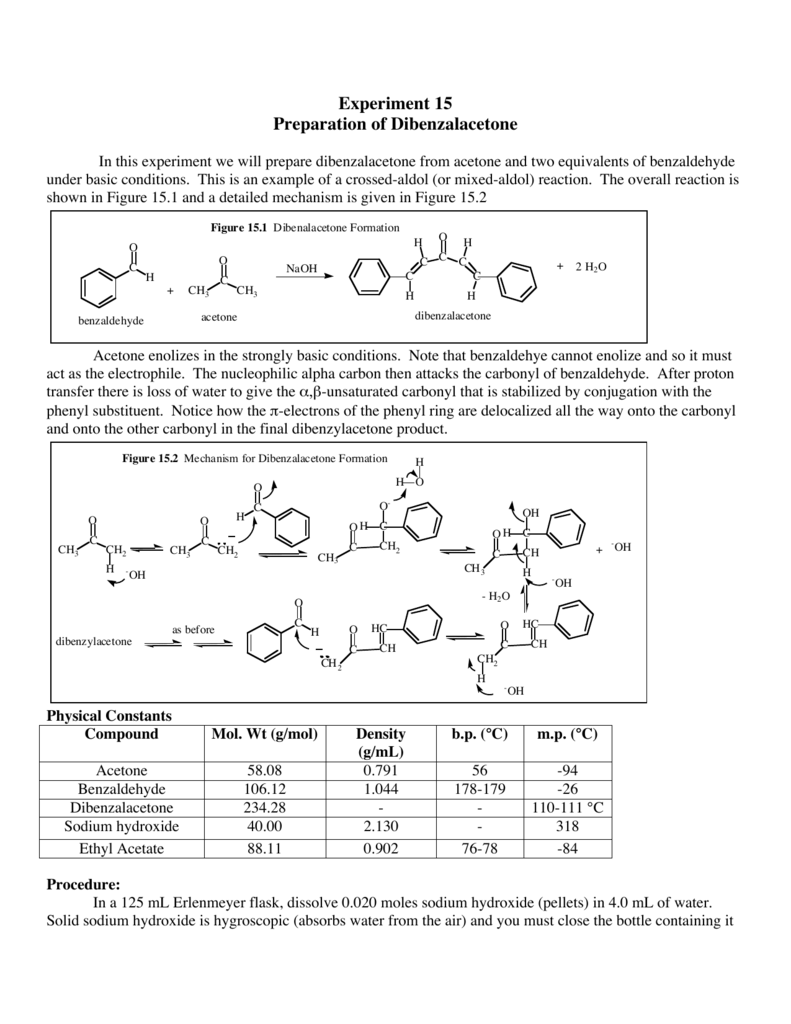
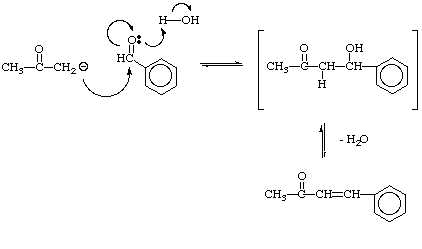
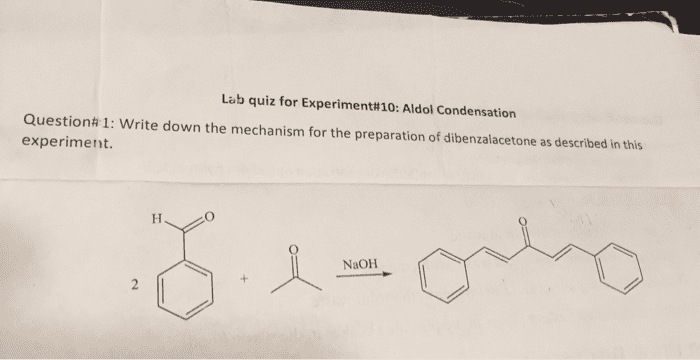
OneClass: Complete and accurate answer to receive points Propose a mechanism for the base-catalyzed c...
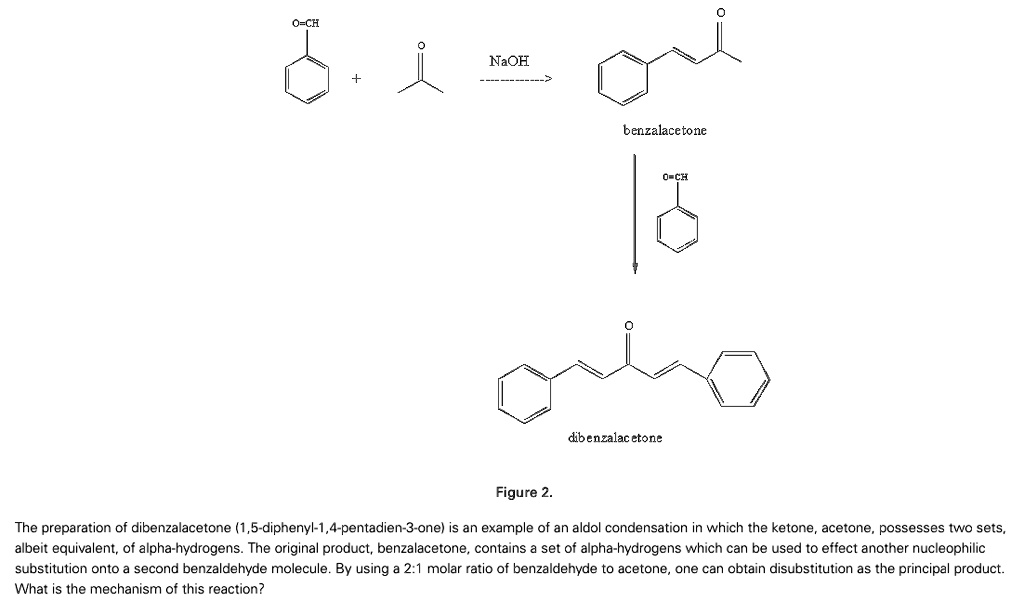
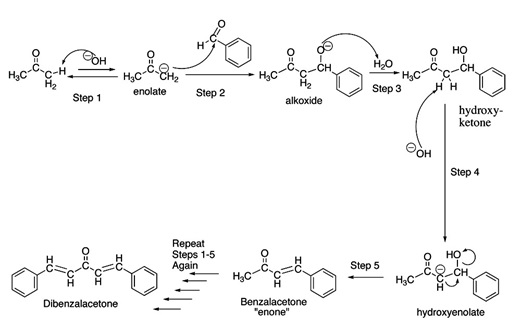
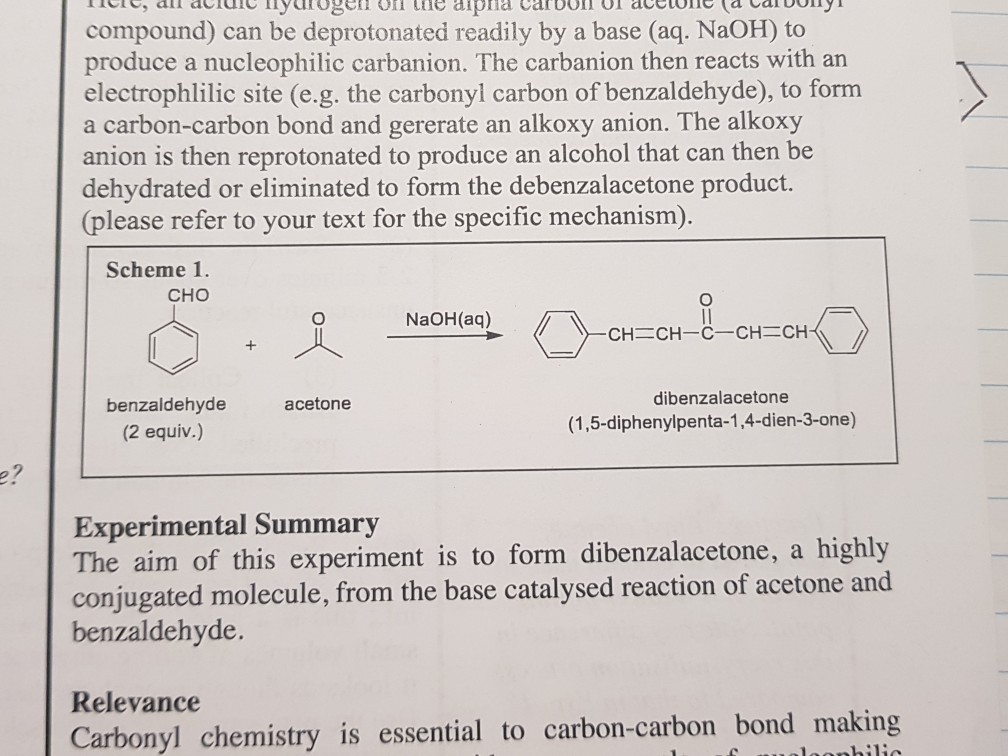
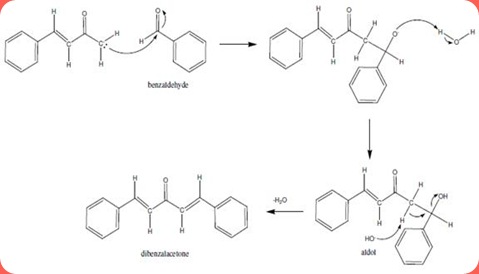
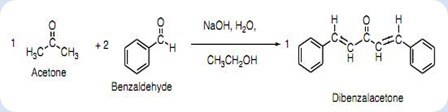
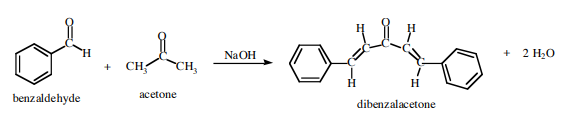
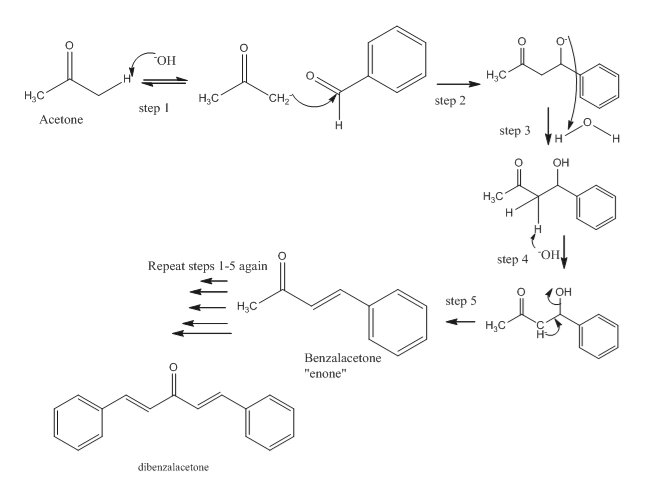
SOLVED:NaOH benzalacetone dibenzalac etone Figure 2 The preparation of dibenzalacetone (1,5-diphenyl-1,4-pentadien-3-one) is an example of an aldol condensation in which the ketone, acetone; possesses two sets albeit equivalent; of alpha-hydrogens. The ...

Lab_8_(Synthesis_of_Dibenzalacetone).docx - University of Missouri - Columbia Lab 8 Synthesis of Dibenzalacetone Worksheet YouTube video | Course Hero
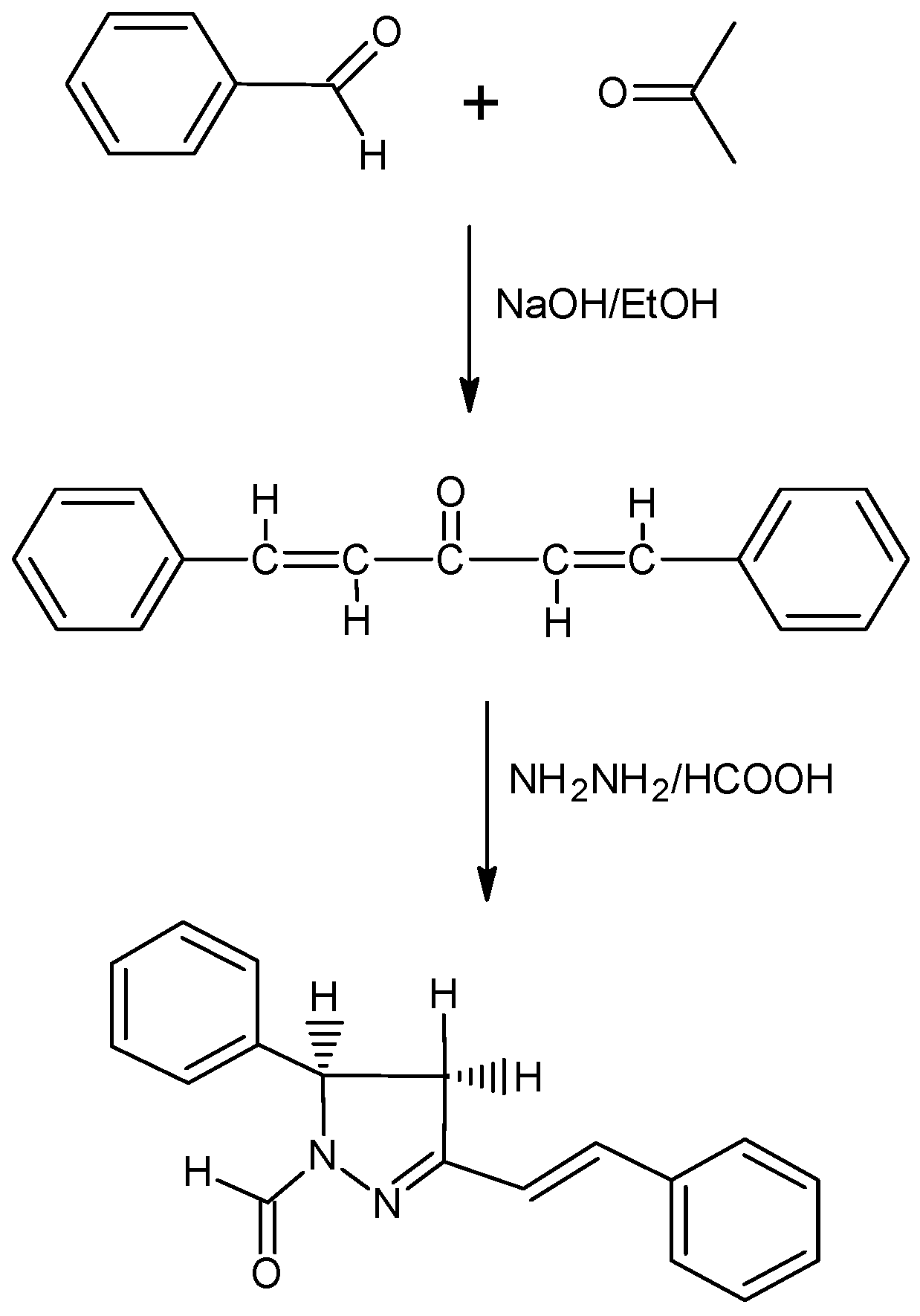
Formation of higher-order structures of chiral poly(ethynylpyridine)s depending on size, temperature, and saccharide recognition - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C2OB25816A

Pori recently did an experiment on Aldol Condensation, by reacting Benzaldehyde with Acetone to form Dibenzalacetone. The proposed stoichiometric ratio of Benzaldehyde: Acetone is 2:1. What would be the implications if either