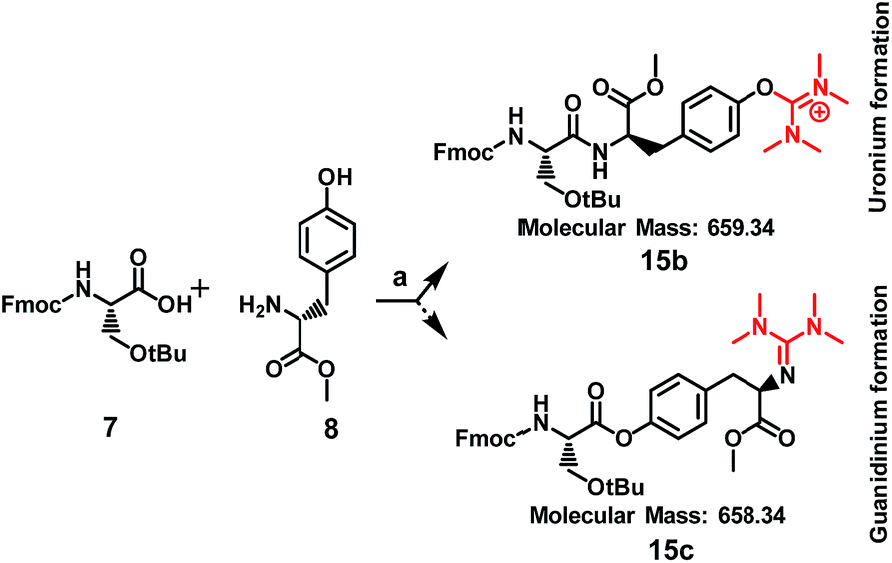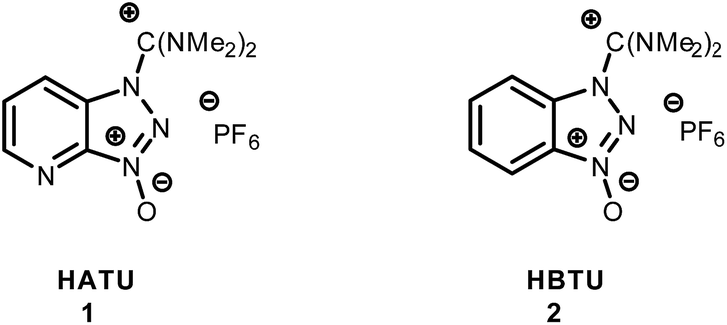
Unveiling and tackling guanidinium peptide coupling reagent side reactions towards the development of peptide-drug conjugates - RSC Advances (RSC Publishing) DOI:10.1039/C7RA06655D
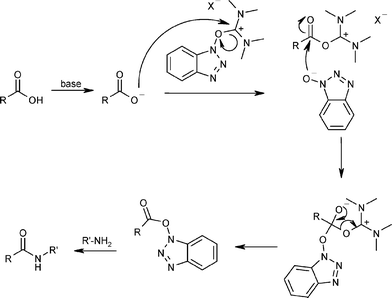
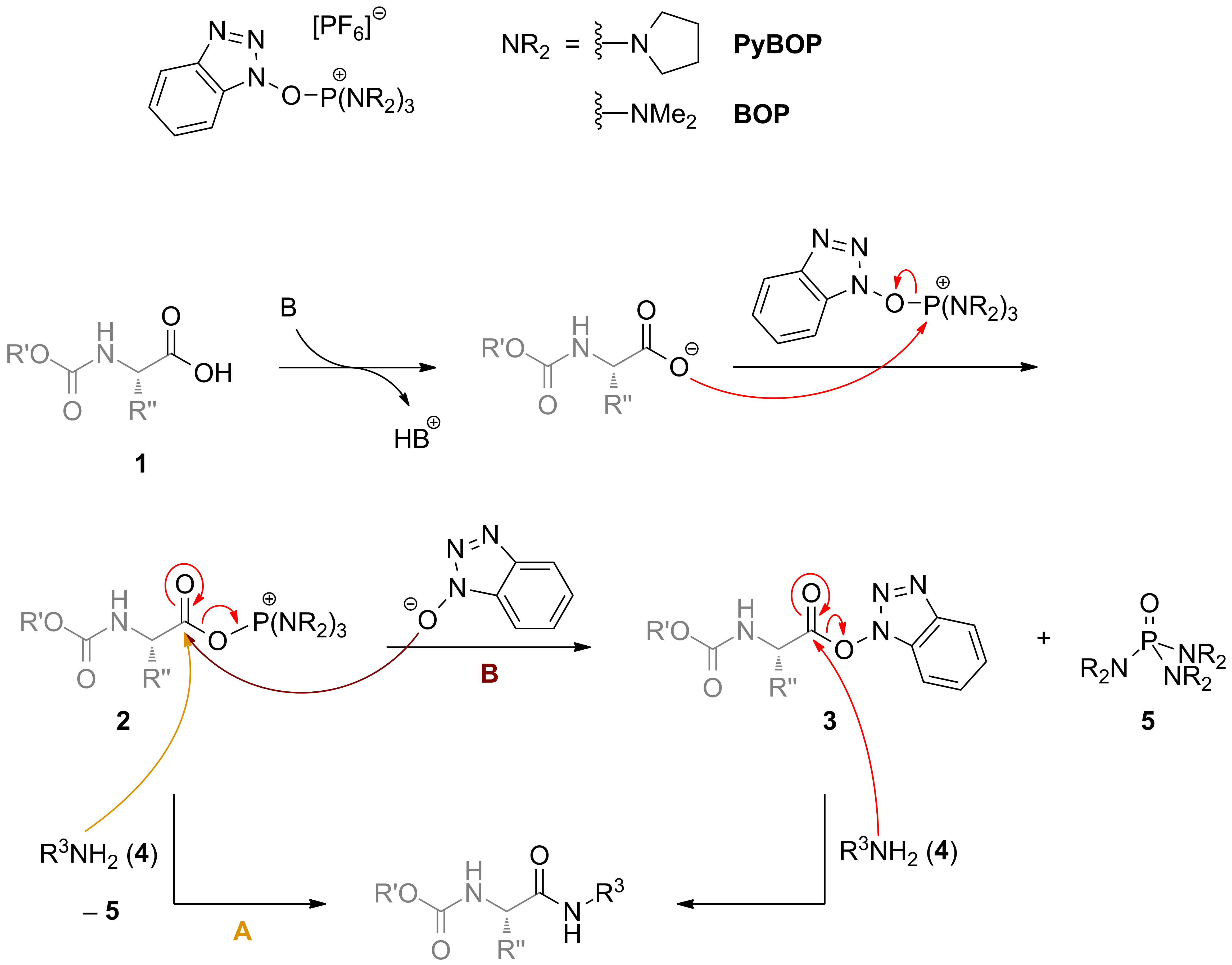
Amide bond formation: beyond the myth of coupling reagents - Chemical Society Reviews (RSC Publishing) DOI:10.1039/B701677H
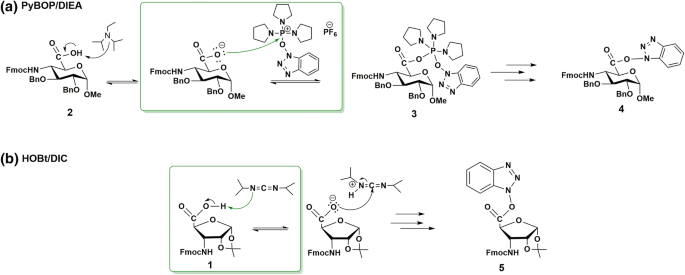
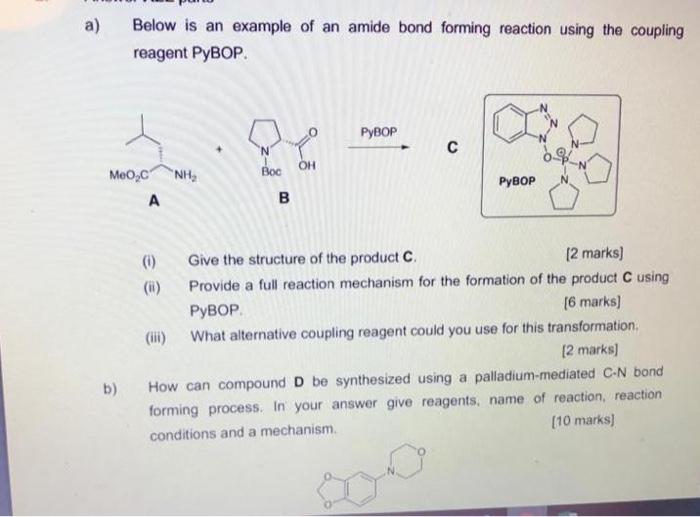
α/β-Chimera peptide synthesis with cyclic β-sugar amino acids: the efficient coupling protocol | SpringerLink

Scheme 16 | Thirteen decades of peptide synthesis: key developments in solid phase peptide synthesis and amide bond formation utilized in peptide ligation | SpringerLink
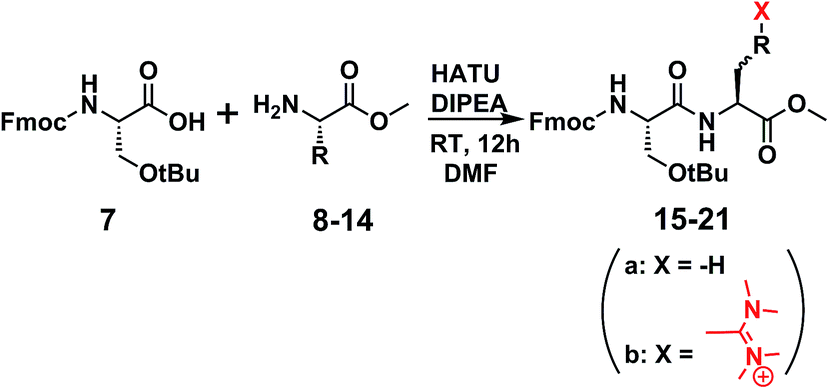
Unveiling and tackling guanidinium peptide coupling reagent side reactions towards the development of peptide-drug conjugates - RSC Advances (RSC Publishing) DOI:10.1039/C7RA06655D
Phosphonium Coupling in the Direct Bond Formations of Tautomerizable Heterocycles via Câ•fiOH Bond Activation<link href=&
Phosphonium Coupling in the Direct Bond Formations of Tautomerizable Heterocycles via Câ•fiOH Bond Activation<link href=&
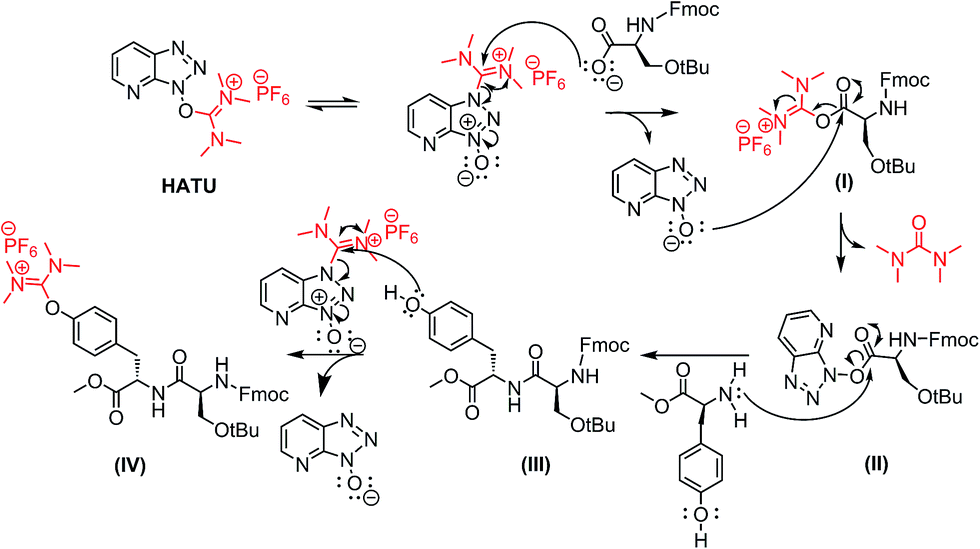
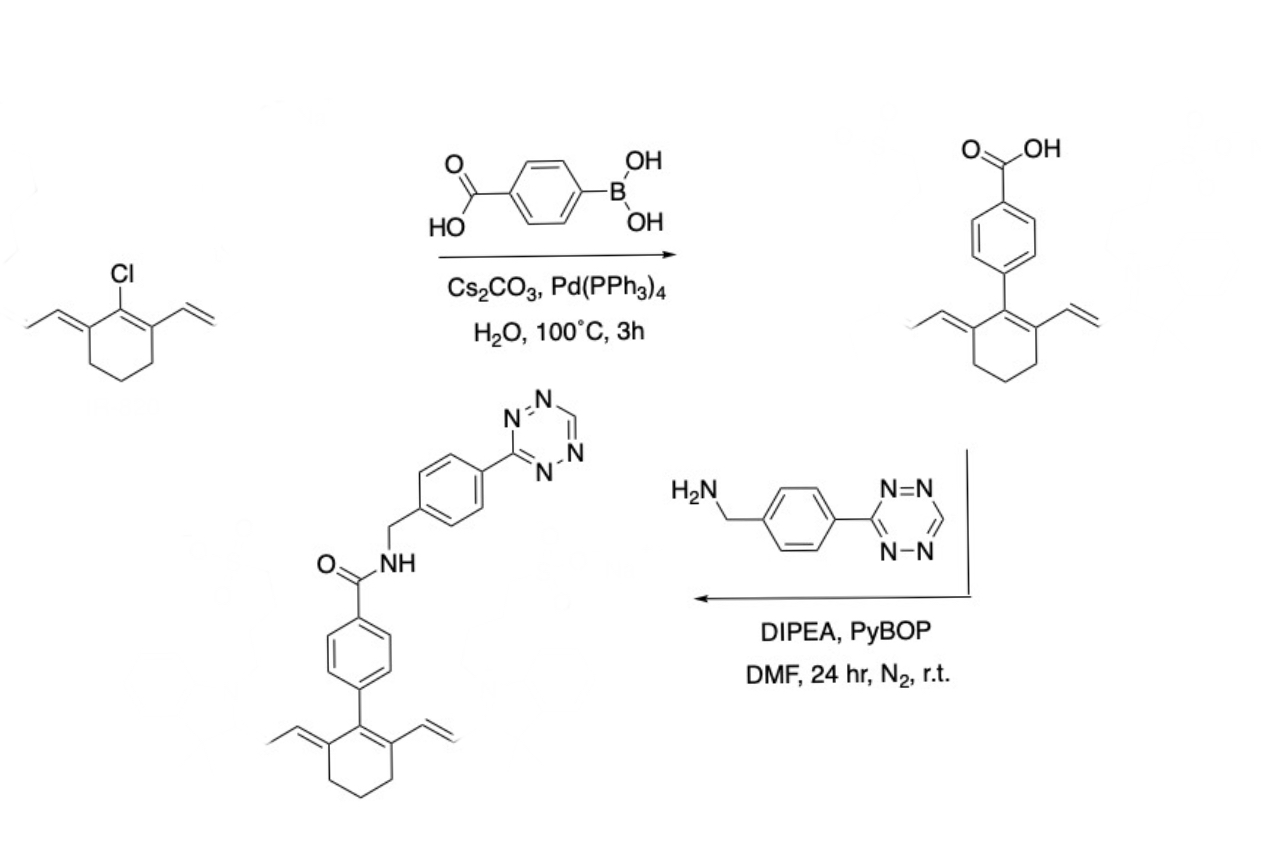
Unveiling and tackling guanidinium peptide coupling reagent side reactions towards the development of peptide-drug conjugates - RSC Advances (RSC Publishing) DOI:10.1039/C7RA06655D