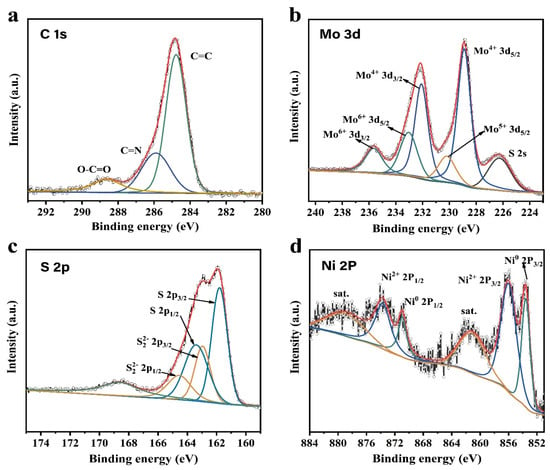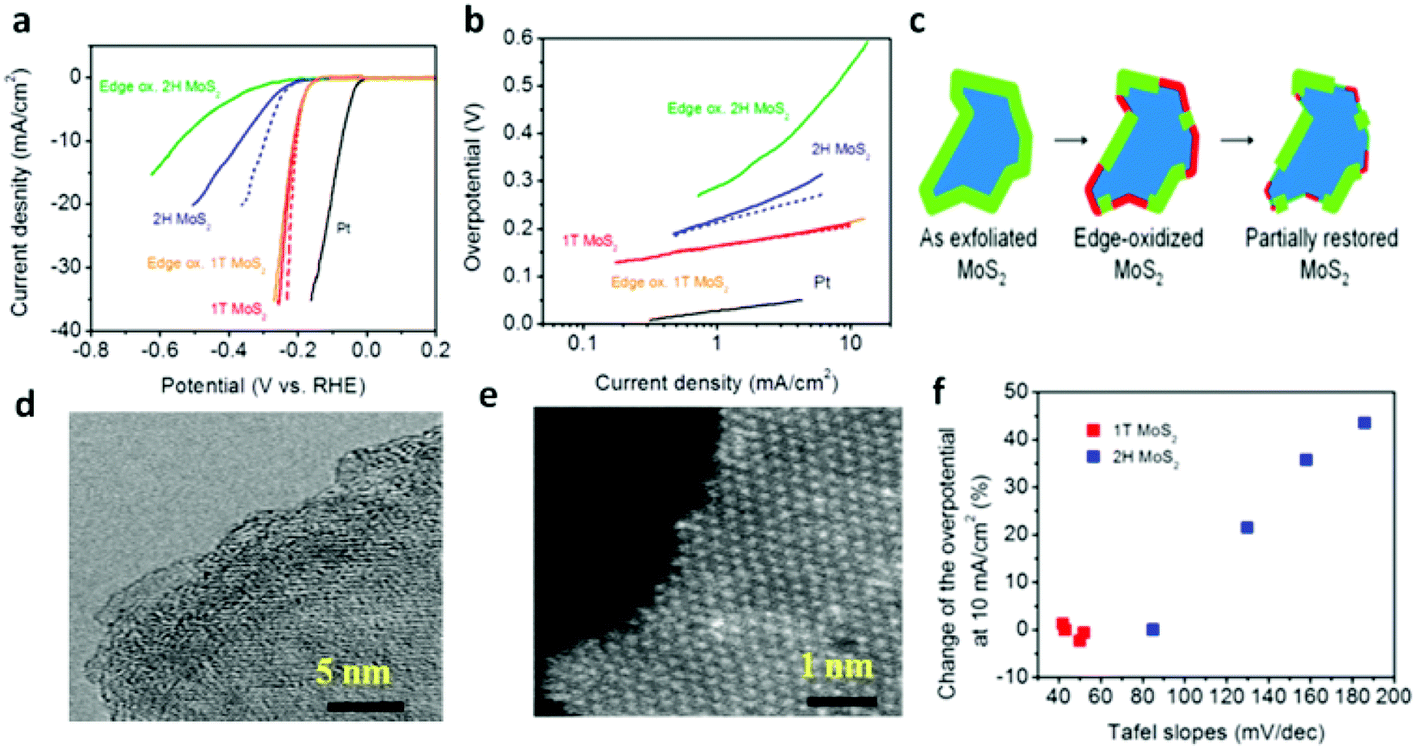
Recent advances in transition-metal dichalcogenide based nanomaterials for water splitting - Nanoscale (RSC Publishing)

Energy‐Saving Electrolytic Hydrogen Generation: Ni2P Nanoarray as a High‐Performance Non‐Noble‐Metal Electrocatalyst - Tang - 2017 - Angewandte Chemie - Wiley Online Library

Influence of Bubbles on the Energy Conversion Efficiency of Electrochemical Reactors - ScienceDirect

Metal–Organic Frameworks Based Electrocatalysts for the Oxygen Reduction Reaction - Lu - 2020 - Angewandte Chemie - Wiley Online Library

Processable Surface Modification of Nickel‐Heteroatom (N, S) Bridge Sites for Promoted Alkaline Hydrogen Evolution - Li - 2019 - Angewandte Chemie - Wiley Online Library
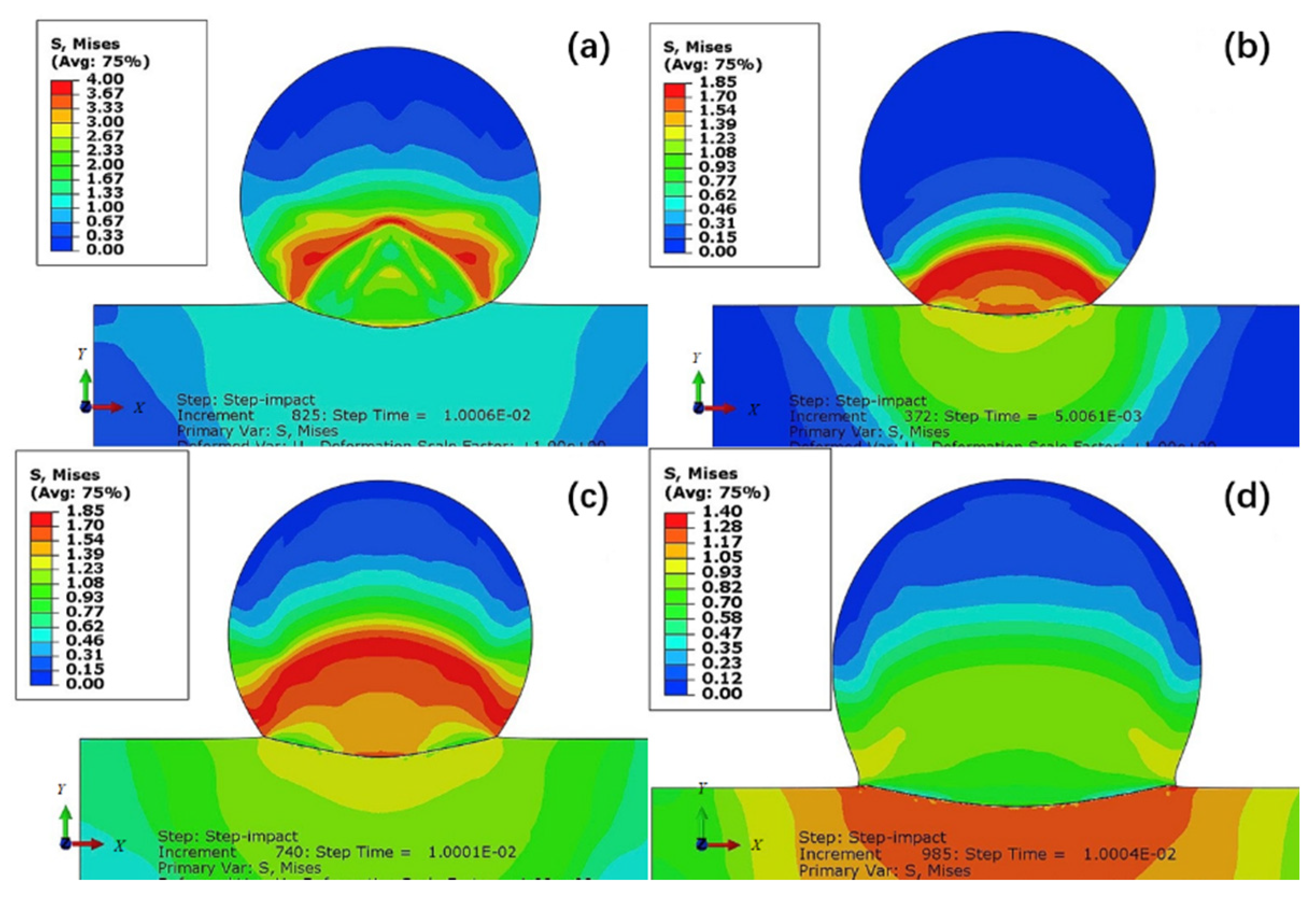
Coatings | Free Full-Text | Current Research Status on Cold Sprayed Amorphous Alloy Coatings: A Review | HTML
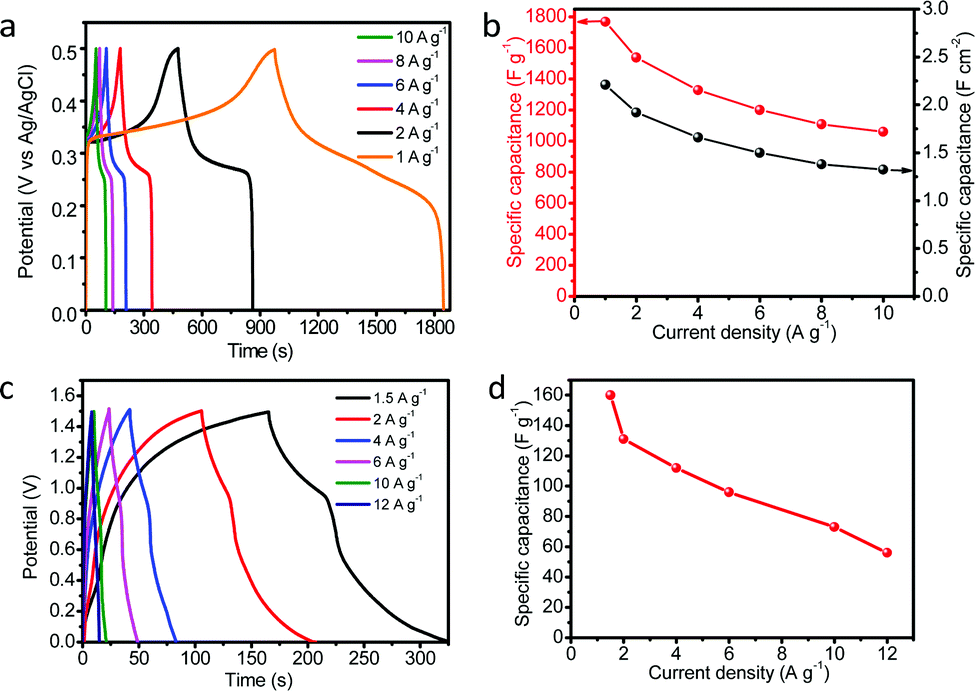
Highly active hollow mesoporous NiFeCr hydroxide as an electrode material for the oxygen evolution reaction and a redox capacitor - Chemical Communications (RSC Publishing)
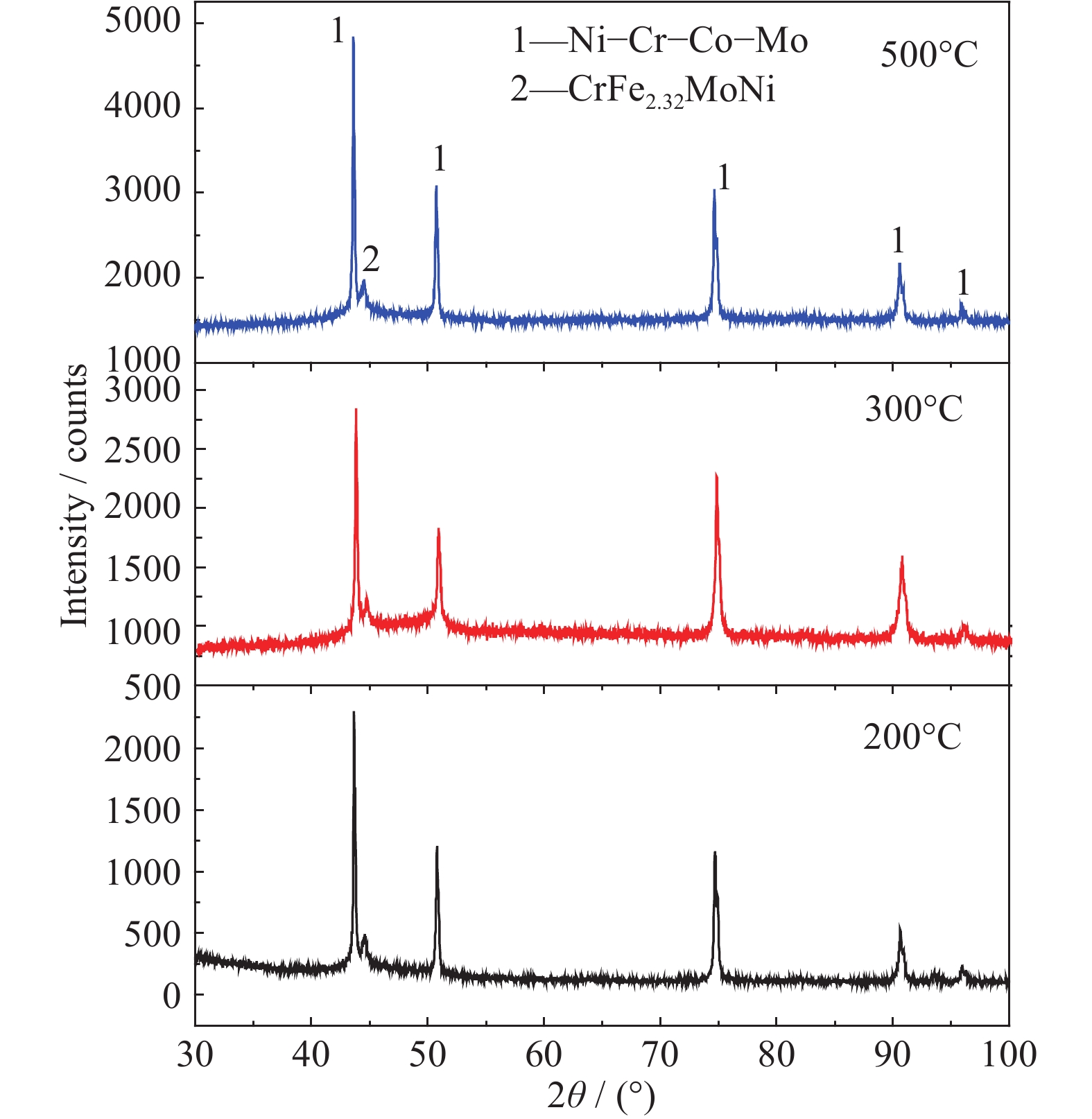
Effects of substrate temperature and deposition time on the morphology and corrosion resistance of FeCoCrNiMo0.3 high-entropy alloy coating fabricated by magnetron sputtering
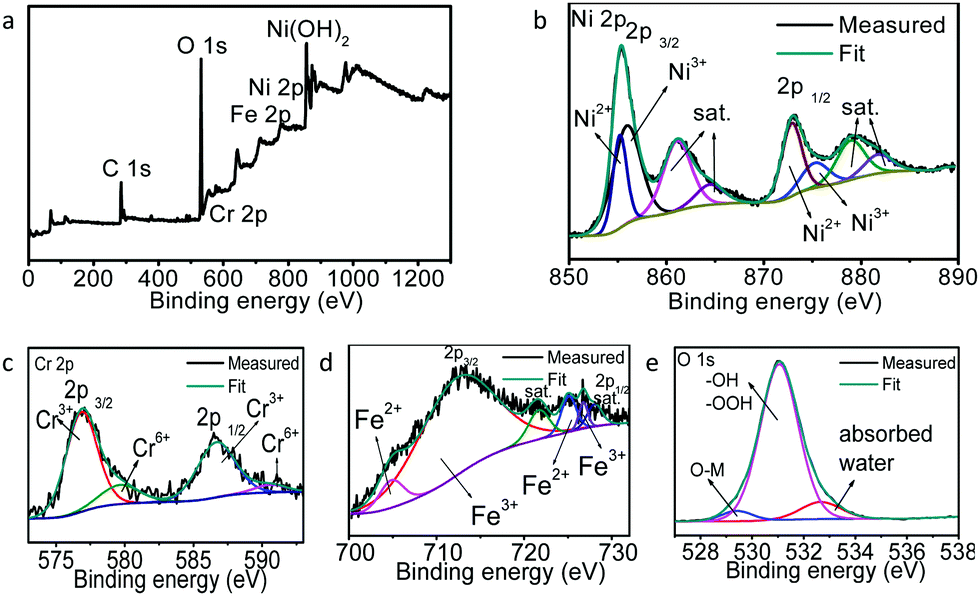
Highly active hollow mesoporous NiFeCr hydroxide as an electrode material for the oxygen evolution reaction and a redox capacitor - Chemical Communications (RSC Publishing)
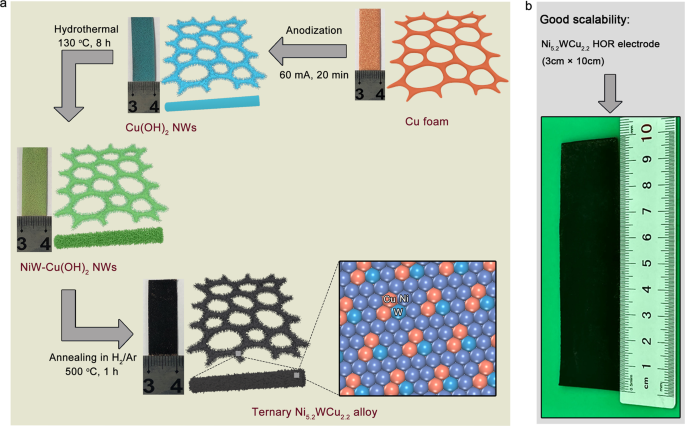
Ternary nickel–tungsten–copper alloy rivals platinum for catalyzing alkaline hydrogen oxidation | Nature Communications

Nickel/Nickel(II) Oxide Nanoparticles Anchored onto Cobalt(IV) Diselenide Nanobelts for the Electrochemical Production of Hydrogen - Xu - 2013 - Angewandte Chemie International Edition - Wiley Online Library