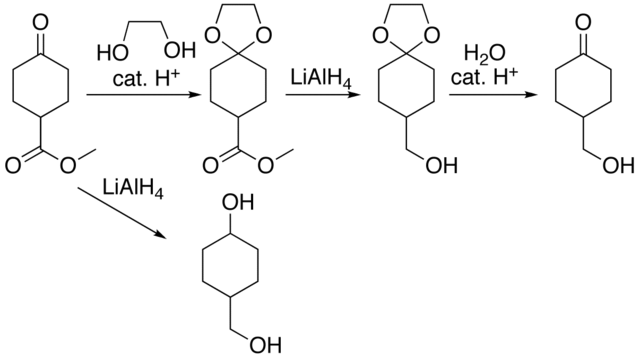
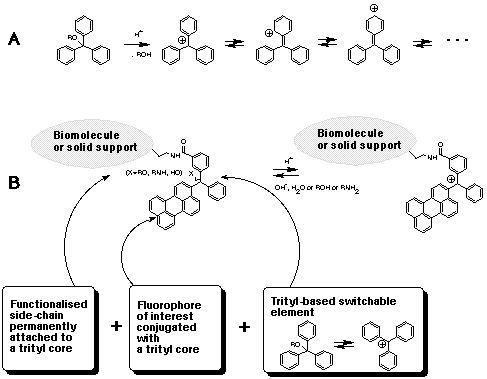
Synthesis of Peptides Containing C-Terminal Esters Using Trityl Side-Chain Anchoring: Applications to the Synthesis of C-Terminal Ester Analogs of the Saccharomyces cerevisiae Mating Pheromone a-Factor. - Abstract - Europe PMC
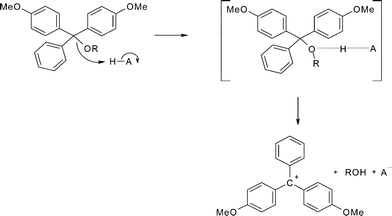
The kinetics and mechanism of the acid-catalysed detritylation of nucleotides in non-aqueous solution - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/B816235B
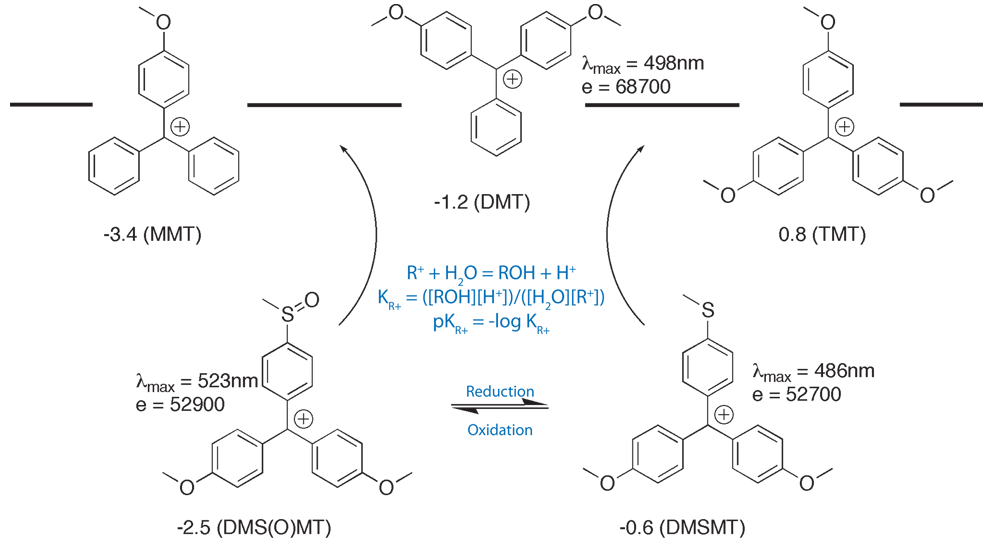
Glen Report 13.13 - Trityl Group in the 3rd Millenium:<br> New Perspectives for Oligonucleotide Chemistry and Beyond
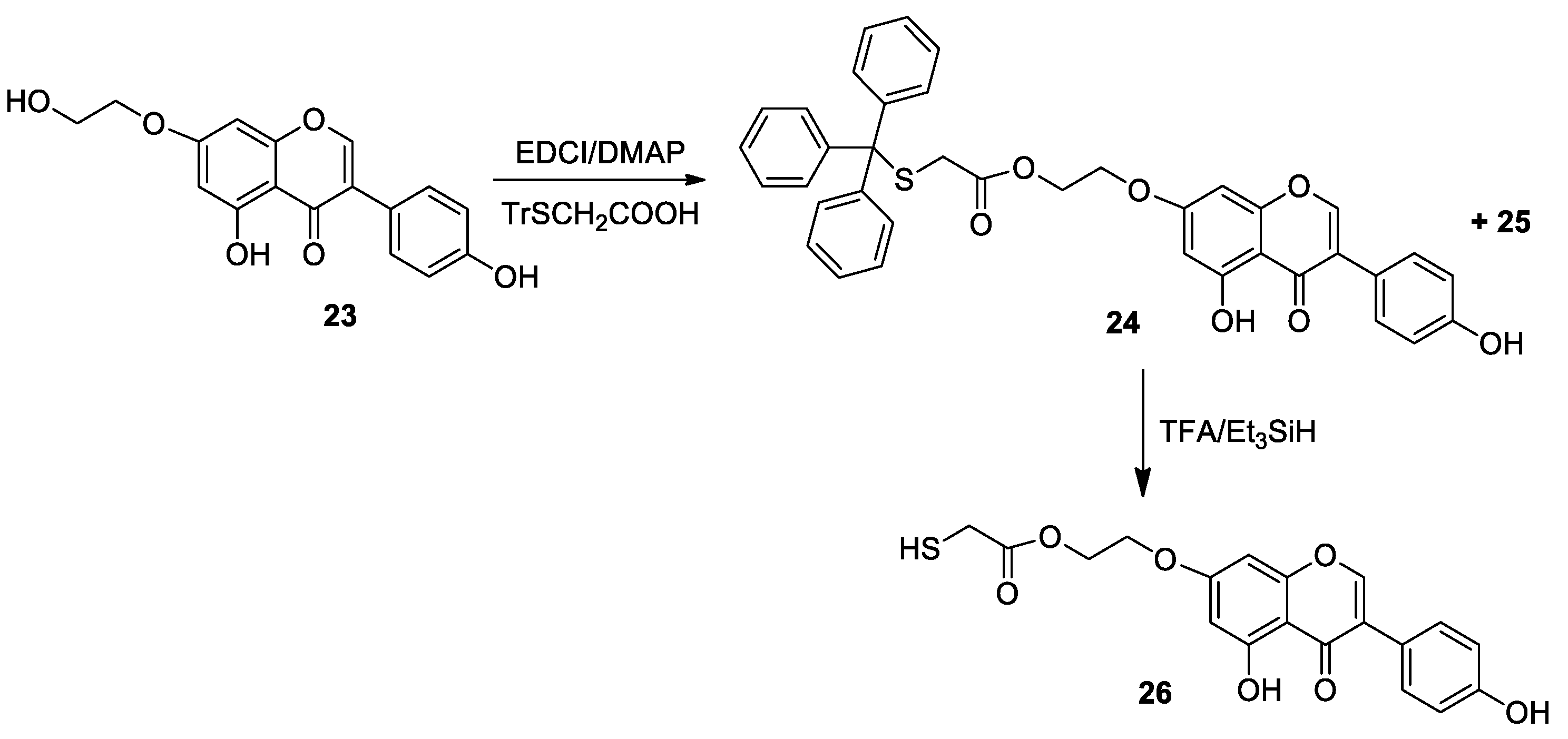
Molecules | Free Full-Text | Synthesis of Thiol Derivatives of Biological Active Compounds for Nanotechnology Application | HTML

Stabilities of Trityl‐Protected Substrates: The Wide Mechanistic Spectrum of Trityl Ester Hydrolyses - Horn - 2010 - Chemistry – A European Journal - Wiley Online Library

Arnold Group Literature Blog: Thorium-ligand multiple bonds via reductive deprotection of a trityl group - Chemical Science (RSC Publishing)
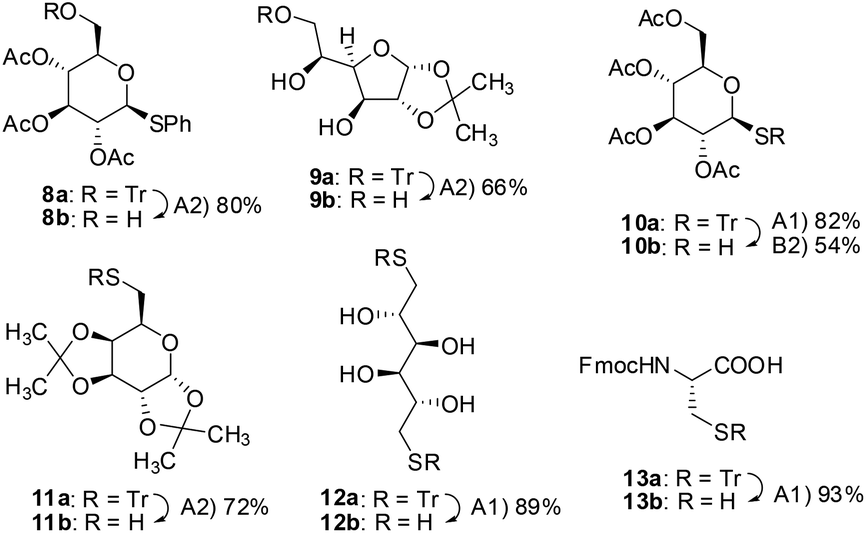
A three-component reagent system for rapid and mild removal of O -, N - and S -trityl protecting groups - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C6OB00067C

LiCl-mediated, easy, and low-cost removal of the trityl group from protected alcohols and diols - ScienceDirect
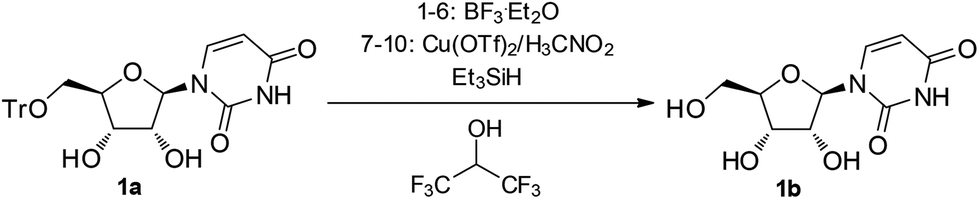
A three-component reagent system for rapid and mild removal of O -, N - and S -trityl protecting groups - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C6OB00067C
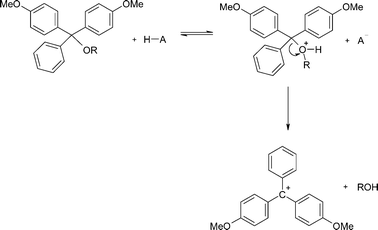
The kinetics and mechanism of the acid-catalysed detritylation of nucleotides in non-aqueous solution - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/B816235B
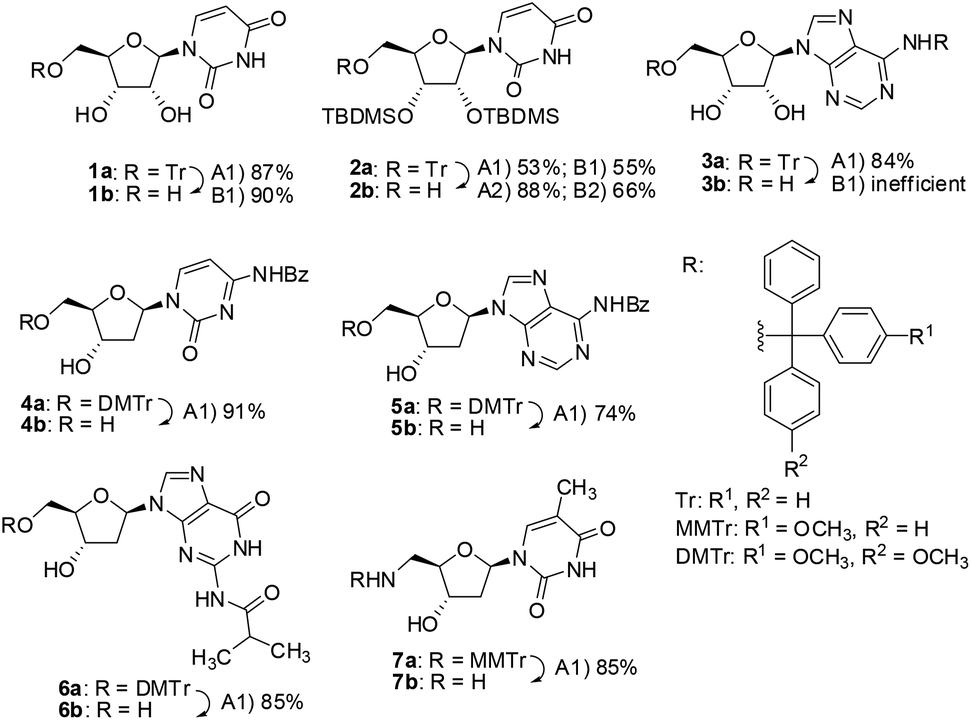
A three-component reagent system for rapid and mild removal of O -, N - and S -trityl protecting groups - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C6OB00067C