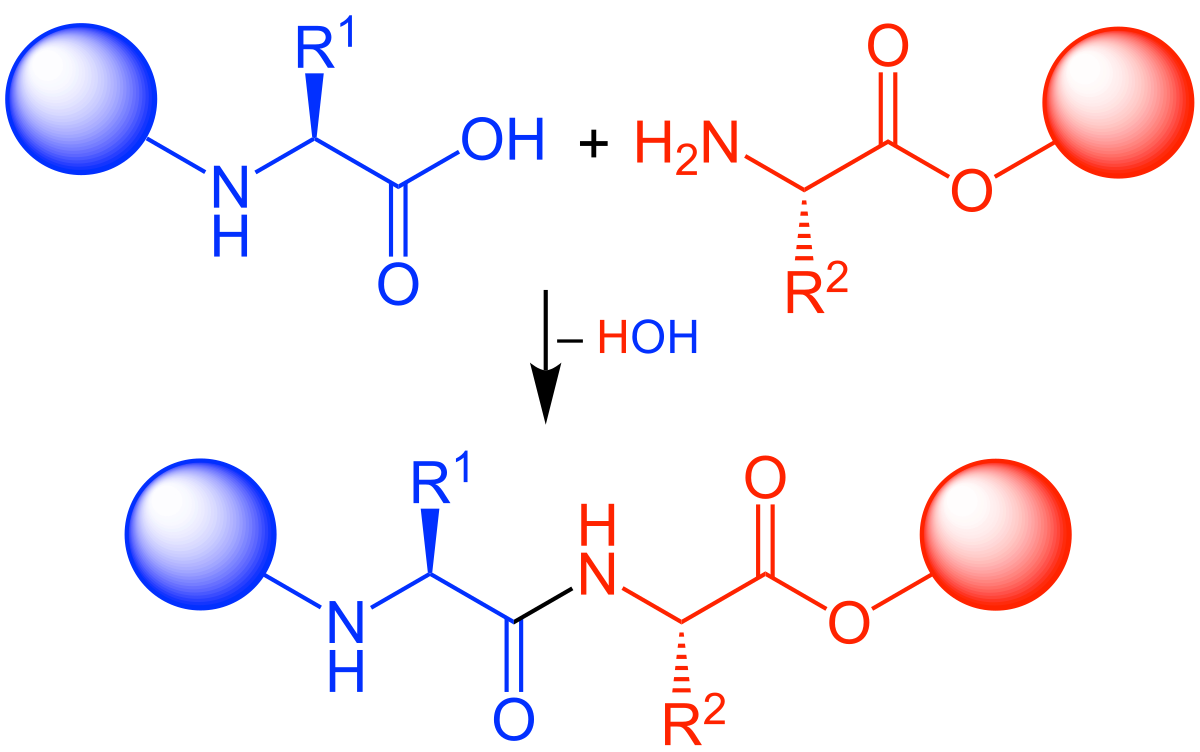
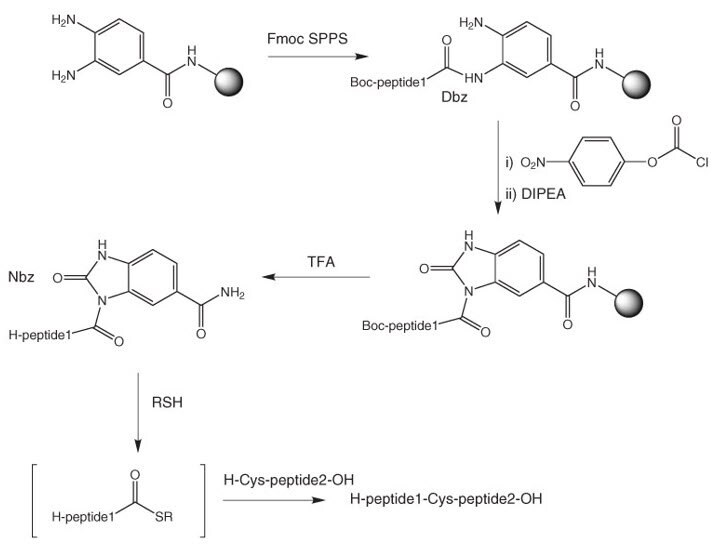
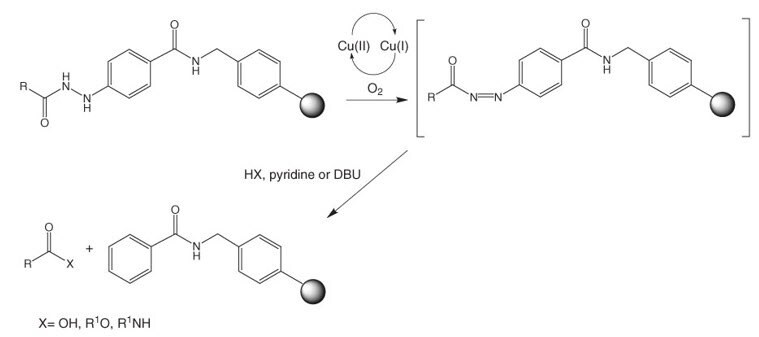
SciELO - Brasil - Highlights in the solid-phase organic synthesis of natural products and analogues Highlights in the solid-phase organic synthesis of natural products and analogues
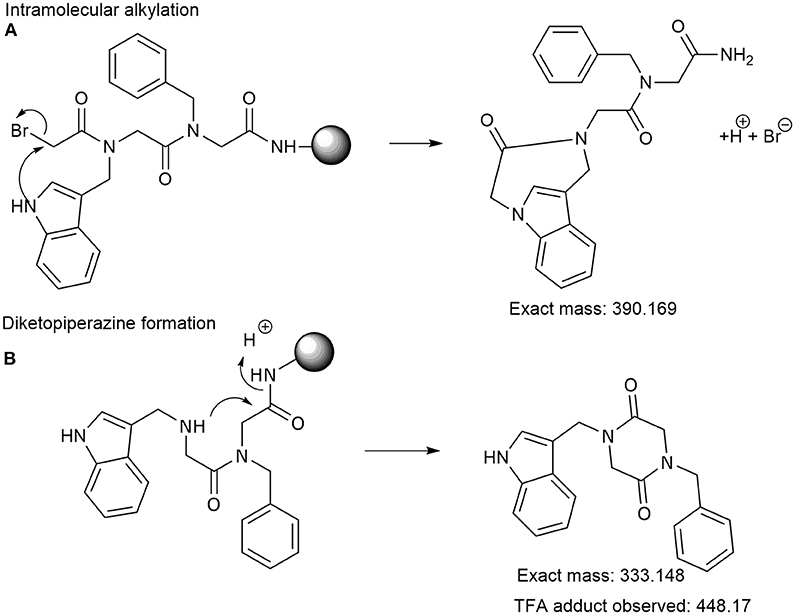
Frontiers | Synthesis of Peptoids Containing Multiple Nhtrp and Ntrp Residues: A Comparative Study of Resin, Cleavage Conditions and Submonomer Protection | Chemistry

On resin click-chemistry-mediated synthesis of novel enkephalin analogues with potent anti-nociceptive activity | Scientific Reports

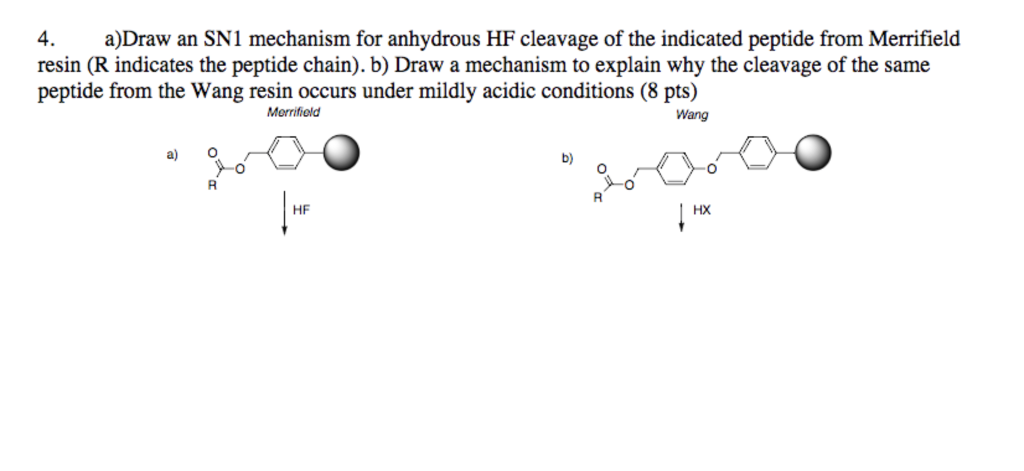
Selective Bi‐directional Amide Bond Cleavage of N‐Methylcysteinyl Peptide - Qiu - 2014 - European Journal of Organic Chemistry - Wiley Online Library
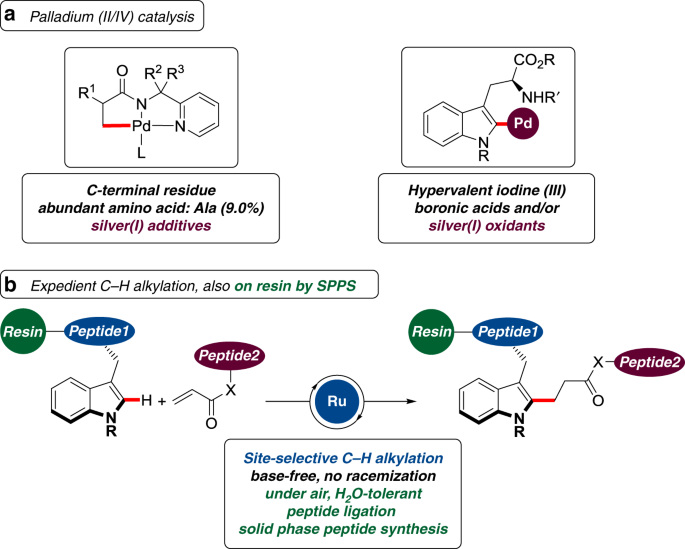
Late-stage peptide C–H alkylation for bioorthogonal C–H activation featuring solid phase peptide synthesis | Nature Communications

SciELO - Brasil - Highlights in the solid-phase organic synthesis of natural products and analogues Highlights in the solid-phase organic synthesis of natural products and analogues
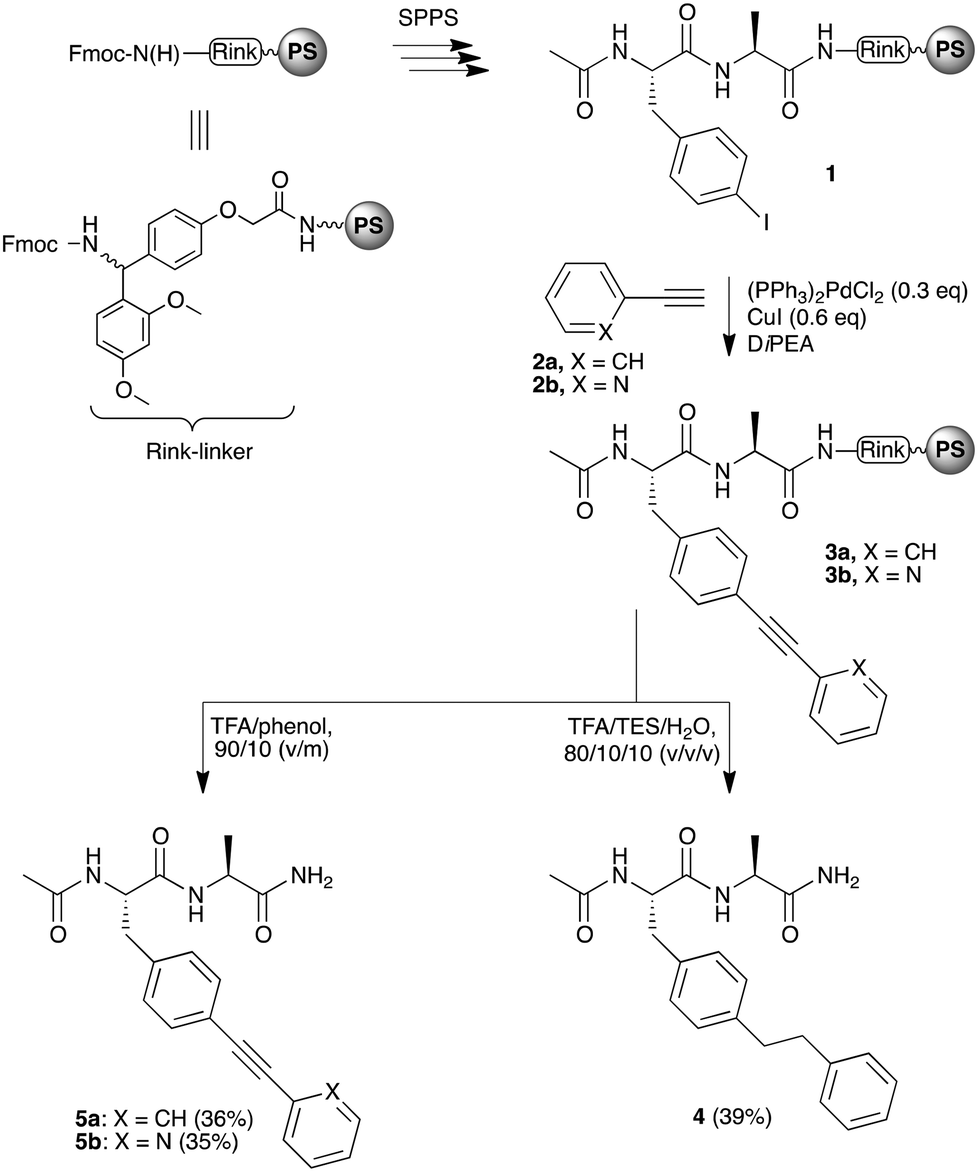
Synthesis of bisarylethyne–peptide conjugates - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/C4QO00357H

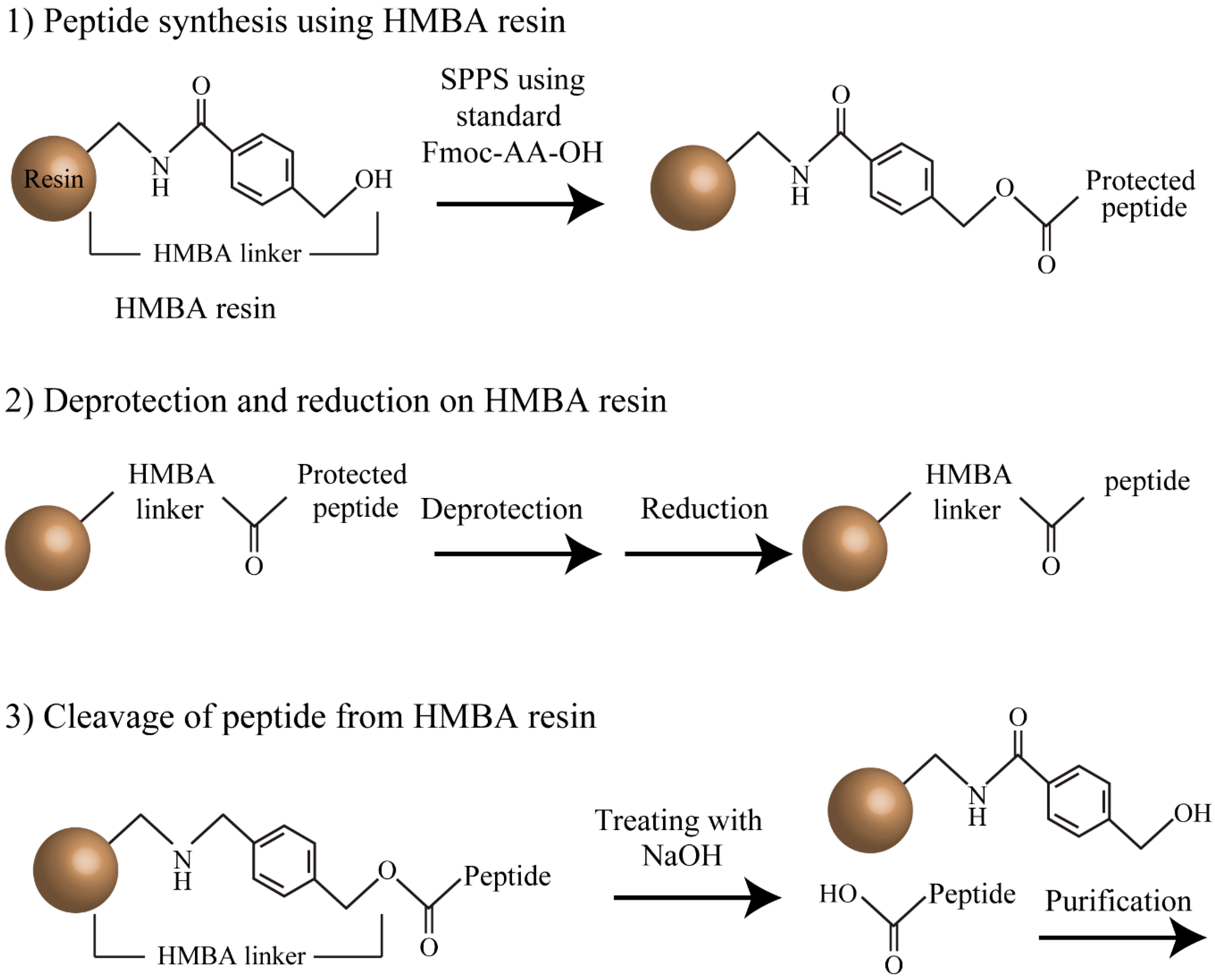
A general solid phase method for the synthesis of depsipeptides - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C2OB26893K

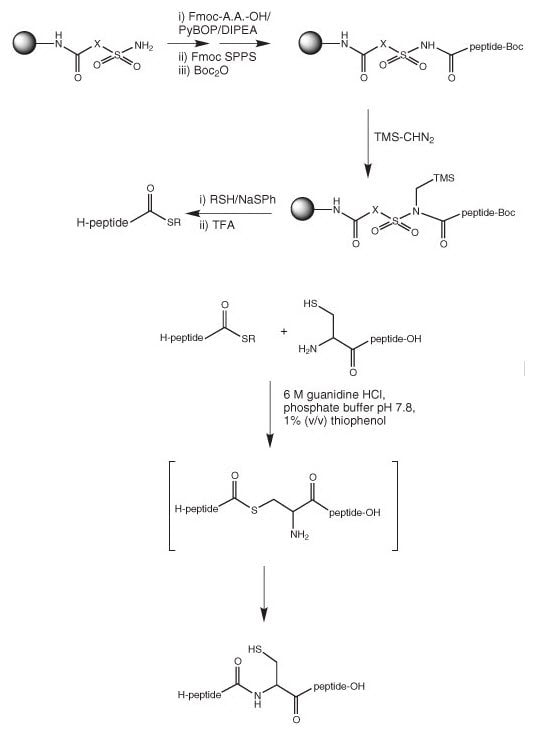
Frontiers | Recent Progress in the Chemical Synthesis of Class II and S-Glycosylated Bacteriocins | Microbiology